-
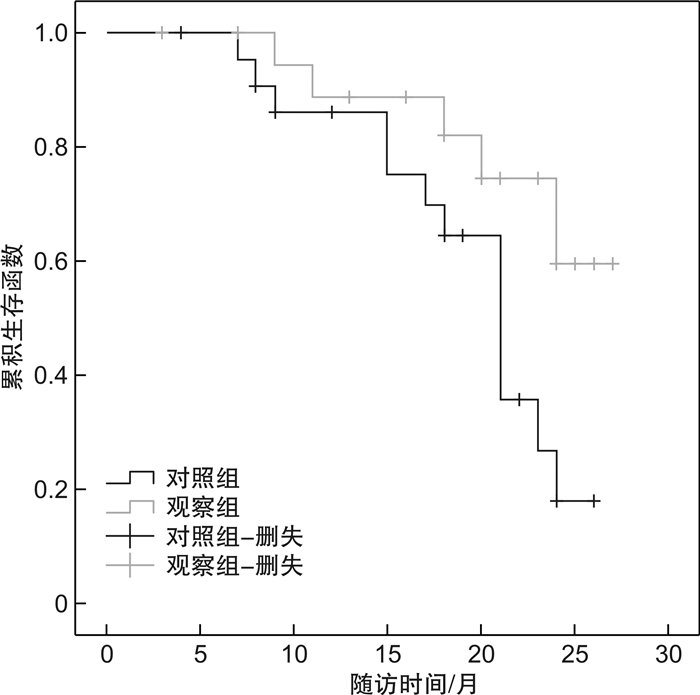
摘要: 目的 探讨PD-1抑制剂联合阿西替尼对转移性肾癌的治疗效果及安全性。方法 纳入2018年1月—2019年4月三明市第二医院收治的转移性肾癌患者43例,根据二线治疗方法的不同,分为对照组和观察组。对照组患者接受阿西替尼治疗,观察组患者在对照组的基础上接受帕博利珠单抗注射液进行治疗。对疾病控制率、客观缓解率进行计算。对患者在治疗期间的毒副反应等级进行记录,分为1~4级,随着等级增加,毒副反应严重程度增加。同时对患者在治疗过程中出现的毒副反应进行记录。结果 治疗后,对照组疾病控制率为69.57%(16/23),客观缓解率为26.09%(6/23),1年生存率为82.61%(19/23),中位无进展生存期(PFS)为23个月,95%置信区间(CI)(18.436~27.564),总生存期(OS)为(19.52±1.28)个月,95%CI(17.019~22.025)。观察组疾病控制率为85.00%(17/20),客观缓解率为35.00%(7/20),1年生存率为90.00%(18/20),中位PFS为24个月,95%CI(22.413~25.587),OS为(23.53±1.38)个月,95%CI(20.819~26.233)。对照组的OS明显低于观察组(t=-9.893,P< 0.01)。腹泻、高血压和乏力均为观察组和对照组出现的主要不良反应。两组毒副反应3级以上发生率比较,差异无统计学意义(P>0.05)。结论 PD-1抑制剂联合阿西替尼治疗转移性肾癌,疾病控制率、PFS、安全性均较好,能够有效提高患者OS,值得在临床中进行推广应用。Abstract: Objective To study the efficacy and safety of PD-1 inhibitor combined with axitinib in the treatment of metastatic renal cell carcinoma.Methods From Jan. 2018 to Apr. 2019, 43 patients with metastatic renal cell carcinoma in Second Hospital of Sanming City were divided into control group and observation group according to different second-line treatment methods. The control group was treated with axitinib, while the observation group was treated with pembrolizumab injection on the basis of the control group. The disease control rate and objective remission rate were calculated. The toxic and side effects of patients during treatment were recorded, which were divided into 1-4 grades. With the increase of grade, the severity of toxic and side effects increased. At the same time, the toxic and side effects of patients in the process of treatment were recorded.Results In the control group, the disease control rate was 69.57% (16/23), the objective remission rate was 26.09% (6/23), 1-year survival rate was 82.61% (19/23), the median PFS was 23 months, 95% confidence interval (18.436-27.564), and the OS was (19.52±1.28) months, 95% confidence interval (17.019-22.025). In the observation group, the disease control rate was 85.00%(17/20), the objective remission rate was 35.00% (7/20), 1-year survival rate was 90.00% (18/20), the median PFS was 24 months, 95% confidence interval (22.413-25.587), and the OS was (23.53±1.38) months, 95% confidence interval (20.819-26.233). The OS of the control group was significantly lower than that of the observation group (t=-9.893,P< 0.01). Diarrhea, hypertension and fatigue were the main adverse reactions in the observation group and the control group. There was no significant difference between the two groups in the incidence of grade 3 or above (P> 0.05).Conclusion PD-1 inhibitor combined with axitinib in the treatment of metastatic renal cell carcinoma has good disease control rate, PFS and safety, and can effectively improve the OS of patients, so it is worthy of clinical application.
-
Key words:
- PD-1 inhibitor /
- axitinib /
- metastatic renal cell carcinoma
-

-
表 表 1 两组基线资料比较
例 组别 例数 性别 中位年龄/岁 转移部位 IMDC危险分层 Fuhrman核分级 ECOG评分 男 女 肺 肝脏 骨 ≥2个部位 低危 中危 高危 Ⅰ级 Ⅱ级 Ⅲ级 Ⅳ级 0分 1分 对照组 23 15 8 63(42~76) 16 4 3 1 5 15 3 7 10 4 2 18 5 观察组 20 13 7 61(43~79) 14 3 3 1 4 14 2 6 9 4 1 14 6 表 表 2 两组疗效比较
组别 例数 CR PR SD PD 疾病控制率 客观缓解率 1年生存率 中位PFS/月 OS/月 对照组 23 0 6 10 7 69.57%(16/23) 26.09%(6/23) 82.61%(19/23) 23 19.52±1.28 观察组 20 0 7 10 3 85.00%(17/20) 35.00%(7/20) 90.00%(18/20) 24 23.53±1.381) 与对照组比较,1)P < 0.05。 表 表 3 两组毒副反应比较
例(%) 毒副反应 观察组(n=20) 对照组(n=23) 总病例数 毒副反应3级以上占比 总病例数 毒副反应3级以上占比 腹泻 11(55.00) 3(15.00) 13(56.52) 2(8.70) 高血压 9(45.00) 4(20.00) 8(34.78) 2(8.70) 乏力 8(40.00) 1(5.00) 9(39.13) 1(4.35) 甲状腺功能减退 6(30.00) 0 5(21.74) 0 食欲减退 7(35.00) 3(15.00) 6(26.09) 2(8.70) 丙氨酸转移酶升高 5(25.00) 2(10.00) 4(17.39) 2(8.70) 天冬氨酸转移酶升高 6(30.00) 1(5.00) 7(30.43) 2(8.70) 蛋白尿 4(20.00) 1(5.00) 3(13.04) 2(8.70) 皮疹 2(10.00) 0 1(4.35) 0 呕吐 3(15.00) 1(5.00) 4(17.39) 1(4.35) 白细胞计数下降 1(5.00) 0 0 0 关节痛 4(20.00) 1(5.00) 3(13.04) 0 -
[1] [2] [3] [4] [5] [6] [7] [8] [9] [10] [11] [12] [13] [14] [15] [16] -

| 引用本文: | 王永富, 唐华, 陈剑, 等. PD-1抑制剂联合阿西替尼对转移性肾癌的治疗效果及安全性分析[J]. 临床泌尿外科杂志, 2022, 37(3): 180-183. doi: 10.13201/j.issn.1001-1420.2022.03.005 |
| Citation: | WANG Yongfu, TANG Hua, CHEN Jian, et al. PD-1 inhibitor combined with axitinib for metastatic renal cell carcinoma[J]. J Clin Urol, 2022, 37(3): 180-183. doi: 10.13201/j.issn.1001-1420.2022.03.005 |
- Figure 图 1.



 下载:
下载: