Prediction value of adverse reactions after BCG for efficacy in patients with non-muscle-invasive bladder cancer
-
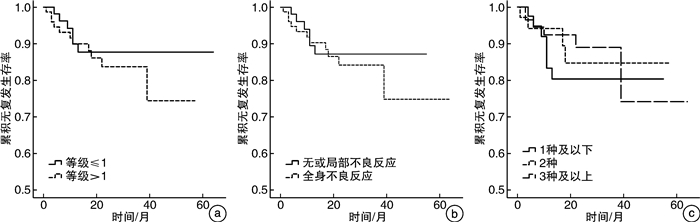
摘要: 目的 探究非肌层浸润性膀胱癌(NMIBC)患者卡介苗(BCG)膀胱灌注后不良反应与疗效的关系。方法 回顾性分析2014年4月—2021年4月长海医院收治的141例接受BCG灌注的中、高危NMIBC患者的临床资料。中位随访时间22(1~64)个月,平均年龄(66.2±10.0)岁,中位年龄67(40~98)岁。所有患者均行经尿道膀胱肿瘤切除术(TURBT),术后进行规律BCG膀胱灌注。将单个不良反应进行单因素Cox回归分析,探究单个不良反应与疗效的关系。根据不良反应发生的分级、部位及数量的不同分别进行log-rank分析,探究不良反应不同亚组对疗效的预测作用。结果 121例(85.8%)患者出现BCG灌注相关不良反应。根据CTCAE 5.0标准分为1级不良反应65例和2级及以上不良反应76例,两组累计复发率分别为9.2%(6/65)和14.5%(11/76),差异无统计学意义(P=0.485)。进一步将不良反应分为无或局部不良反应62例和全身不良反应79例,两组累计复发率分别为9.7%(6/62)和13.9%(11/79),差异无统计学意义(P=0.575)。根据不良反应数量分为1种及以下不良反应47例、2种不良反应35例、3种及以上不良反应59例,三组累计复发率分别为14.9%(7/47)、11.4%(4/35)和10.2%(6/59),差异无统计学意义(P=0.705)。结论 BCG灌注引起不良反应的分级,部位及数量均与灌注疗效无显著关联。仍需前瞻性、大样本量研究以进一步明确BCG灌注后不良反应与其疗效的关系。Abstract: Objective To investigate the relationship between adverse reactions and efficacy after bacillus calmette-guerin (BCG) in patients with non-muscle-invasive bladder cancer (NMIBC).Methods The clinical data of 141 patients with middle or high risk NMIBC who received BCG intravesical instillation in Changhai Hospital from April 2014 to April 2021 were retrospectively analyzed. The median time of follow-up was 22 (1-64) months. Patients' mean age was (66.2±10.0) years, and the median age was 67 (40-98) years. All patients underwent transurethral resection of bladder tumor (TURBT), after which BCG instillation was performed regularly. Single adverse reactions were analyzed by univariate Cox regression analysis to explore the relationship between single adverse reaction and efficacy. Log-rank analysis was performed according to the grade, location and number of adverse reactions, respectively, to explore the predictive value of different subgroups of adverse reactions on efficacy.Results BCG related adverse reactions were found in 121 patients (85.8%). According to CTCAE 5.0 standard, there were 65 cases of grade 1 adverse reactions and 76 cases of grade 2 or above. The cumulative recurrence rates of the two groups were 9.2% (6/65) and 14.5% (11/76), respectively, and the difference was not statistically significant (P=0.485). Adverse reactions were further divided into 62 cases with no or local adverse reactions and 79 cases with systemic adverse reactions. The cumulative recurrence rates of the two groups were 9.7% (6/62) and 13.9% (11/79), respectively, with no statistically significant difference (P=0.575). According to the number of adverse reactions, there were 47 cases of 1 type or less adverse reactions, 35 cases of 2 type adverse reactions, and 59 cases of 3 type or more adverse reactions. The cumulative recurrence rates of the three groups were 14.9% (7/47), 11.4% (4/35) and 10.2% (6/59), respectively, and the difference was not statistically significant (P=0.705).Conclusion The grade, location and number of adverse effects after BCG intravesical instillation was not correlated with the efficacy of BCG treatment. Prospective, large sample size studies are needed to further clarify the relationship between BCG post-instillation adverse reactions and efficacy.
-
Key words:
- bladder cancer /
- bacillus calmette-guerin /
- adverse reactions /
- curative effect /
- predictive value
-

-
表 1 不良反应汇总表
不良反应分级 部位 例数(%)a) HR(95%CI) P值 1级 尿频、尿急、尿痛 局部 111(78.7) 1.359(0.390~4.737) 0.630 血尿 局部 58(41.1) 0.460(0.150~1.412) 0.175 排尿困难 局部 20(14.2) / / 关节疼痛 全身 25(17.7) / / 腹泻 全身 2(1.4) / / 头痛 全身 1(0.7) / / 肛门刺激 全身 1(0.7) / / 呕吐 全身 1(0.7) / / 食欲下降 全身 2(1.4) / / 2级 发热 全身 73(51.8) 1.535(0.567~4.154) 0.399 白细胞升高(尿路感染) 局部 4(2.8) / / 结核活动性膀胱炎 全身 1(0.7) / / 荨麻疹 全身 1(0.7) / / 3级 昏倒 全身 1(0.7) / / 肺部结节 全身 1(0.7) / / 阴囊结核 全身 1(0.7) / / 注:a)本表反映单个不良反应的发生情况,同时发生多种不良反应的患者每种不良反应之间互不影响,均统计在内。 -
[1] Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249. doi: 10.3322/caac.21660
[2] Babjuk M, Burger M, Capoun O, et al. European Association of Urology Guidelines on Non-muscle-invasive Bladder Cancer(Ta, T1, and Carcinoma in Situ)[J]. Eur Urol, 2022, 81(1): 75-94. doi: 10.1016/j.eururo.2021.08.010
[3] Chang SS, Boorjian SA, Chou R, et al. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline[J]. J Urol, 2016, 196(4): 1021-1029. doi: 10.1016/j.juro.2016.06.049
[4] Shelley MD, Kynaston H, Court J, et al. A systematic review of intravesical bacillus Calmette-Guérin plus transurethral resection vs transurethral resection alone in Ta and T1 bladder cancer[J]. BJU Int, 2001, 88(3): 209-216. doi: 10.1046/j.1464-410x.2001.02306.x
[5] Lu JL, Xia QD, Lu YH, et al. Efficacy of intravesical therapies on the prevention of recurrence and progression of non-muscle-invasive bladder cancer: A systematic review and network meta-analysis[J]. Cancer Med, 2020, 9(21): 7800-7809. doi: 10.1002/cam4.3513
[6] Porena M, Del Zingaro M, Lazzeri M, et al. Bacillus Calmette-Guérin versus gemcitabine for intravesical therapy in high-risk superficial bladder cancer: a randomised prospective study[J]. Urol Int, 2010, 84(1): 23-27. doi: 10.1159/000273461
[7] Duchek M, Johansson R, Jahnson S, et al. Bacillus Calmette-Guérin is superior to a combination of epirubicin and interferon-alpha2b in the intravesical treatment of patients with stage T1 urinary bladder cancer. A prospective, randomized, Nordic study[J]. Eur Urol, 2010, 57(1): 25-31. doi: 10.1016/j.eururo.2009.09.038
[8] Brausi M, Oddens J, Sylvester R, et al. Side effects of Bacillus Calmette-Guérin(BCG)in the treatment of intermediate-and high-risk Ta, T1 papillary carcinoma of the bladder: results of the EORTC genito-urinary cancers group randomised phase 3 study comparing one-third dose with full dose and 1 year with 3 years of maintenance BCG[J]. Eur Urol, 2014, 65(1): 69-76. doi: 10.1016/j.eururo.2013.07.021
[9] Nummi A, Järvinen R, Sairanen J, et al. A retrospective study on tolerability and complications of bacillus Calmette-Guérin(BCG)instillations for non-muscle-invasive bladder cancer[J]. Scand J Urol, 2019, 53(2-3): 116-122. doi: 10.1080/21681805.2019.1609080
[10] Suzuki S, Shinohara N, Harabayashi T, et al. Complications of bacillus Calmette-Guerin therapy in superficial urothelial cancer: clinical analysis and implications[J]. Int J Clin Oncol, 2002, 7(5): 289-293. doi: 10.1007/s101470200042
[11] Sylvester RJ, van der Meijden AP, Oosterlinck W, et al. The side effects of Bacillus Calmette-Guerin in the treatment of TaT1bladder cancer do not predict its efficacy: results from a European Organisation for Research and Treatment of Cancer Genito-Urinary Group Phase Ⅲ Trial[J]. Eur Urol, 2003, 44(4): 423-428. doi: 10.1016/S0302-2838(03)00371-3
[12] 郭应禄, 那彦群, 叶章群等. 中国泌尿外科和男科疾病诊断治疗指南[M]. 2019版. 北京: 科学出版社, 2019: 44-45.
[13] Morales A, Eidinger D, Bruce AW. Intracavitary Bacillus Calmette-Guerin in the treatment of superficial bladder tumors[J]. J Urol, 1976, 116(2): 180-183. doi: 10.1016/S0022-5347(17)58737-6
[14] Krajewski W, Matuszewski M, Poletajew S, et al. Are There Differences in Toxicity and Efficacy between Various Bacillus Calmette-Guerin Strains in Bladder Cancer Patients? Analysis of 844 Patients[J]. Urol Int, 2018, 101(3): 277-284. doi: 10.1159/000492722
[15] Huang Z, Liu H, Wang Y, et al. Determining optimal maintenance schedules for adjuvant intravesical bacillus Calmette-Guerin immunotherapy in non-muscle-invasive bladder cancer: a systematic review and network meta-analysis[J]. Curr Med Res Opin, 2017, 33(8): 1379-1387. doi: 10.1080/03007995.2017.1326889
[16] Zeng S, Yu X, Ma C, et al. Low-Dose Versus Standard Dose of Bacillus Calmette-Guerin in the Treatment of Nonmuscle Invasive Bladder Cancer: A Systematic Review and Meta-Analysis[J]. Medicine(Baltimore), 2015, 94(49): e2176.
[17] Quhal F, Pradere B, Laukhtina E, et al. Prognostic value of albumin to globulin ratio in non-muscle-invasive bladder cancer[J]. World J Urol, 2021, 39(9): 3345-3352. doi: 10.1007/s00345-020-03586-1
[18] Ferro M, Di Mauro M, Cimino S, et al. Systemic combining inflammatory score(SCIS): a new score for prediction of oncologic outcomes in patients with high-risk non-muscle-invasive urothelial bladder cancer[J]. Transl Androl Urol, 2021, 10(2): 626-635. doi: 10.21037/tau-20-1272
[19] Antonucci M, Defidio L, De Dominicis M, et al. Utility of Preoperative Neutrophil/Lymphocyte Ratio as a New Objective Prognostic Tool in Endoscopically Treated Upper Tract Urothelial Carcinoma: A Retrospective Evaluation[J]. J Endourol, 2020, 34(9): 993-1000. doi: 10.1089/end.2020.0283
[20] Kamat AM, Briggman J, Urbauer DL, et al. Cytokine Panel for Response to Intravesical Therapy(CyPRIT): Nomogram of Changes in Urinary Cytokine Levels Predicts Patient Response to Bacillus Calmette-Guérin[J]. Eur Urol, 2016, 69(2): 197-200. doi: 10.1016/j.eururo.2015.06.023
[21] Kamat AM, Li R, O'Donnell MA, et al. Predicting Response to Intravesical Bacillus Calmette-Guérin Immunotherapy: Are We There Yet? A Systematic Review[J]. Eur Urol, 2018, 73(5): 738-748. doi: 10.1016/j.eururo.2017.10.003
[22] Blanchet P, Droupy S, Eschwege P, et al. Prospective evaluation of Ki-67 labeling in predicting the recurrence and progression of superficial bladder transitional cell carcinoma[J]. Eur Urol, 2001, 40(2): 169-175. doi: 10.1159/000049768
[23] Langle YV, Belgorosky D, Prack McCormick B, et al. FGFR3 Down-Regulation is Involved in bacillus Calmette-Guérin Induced Bladder Tumor Growth Inhibition[J]. J Urol, 2016, 195(1): 188-197. doi: 10.1016/j.juro.2015.06.093
-




 下载:
下载: