-
摘要: 继发性高血压最常见的肾上腺疾病是嗜铬细胞瘤/副神经节瘤(PPGL)和原发性醛固酮增多症(PA),尤其是PPGL。PPGL大多在病理上是良性,但生物学行为具有恶性潜能,有因心、脑、血管并发症或急剧发作危象而死亡的危险,因此早期诊断尤其重要。本文综述了近几年PPGL的诊断进展,旨在帮助临床医师早期诊断,以指导治疗和改善预后。Abstract: The most common adrenal diseases of secondary hypertension are pheochromocytoma/paraganglioma(PPGL) and primary aldosteronism (PA), especially PPGL. Although most PPGL is pathologically benign, its biological behavior has malignant potential, and there is a risk of death due to cardiac, cerebral, vascular complications or acute crisis. Therefore, early diagnosis is important especially. In this review, the diagnostic progress of PPGL in recent years was reviewed to help clinicians to make early diagnosis to guide treatment and improve prognosis.
-
Key words:
- adrenal diseases /
- pheochromocytoma /
- diagnosis
-

-
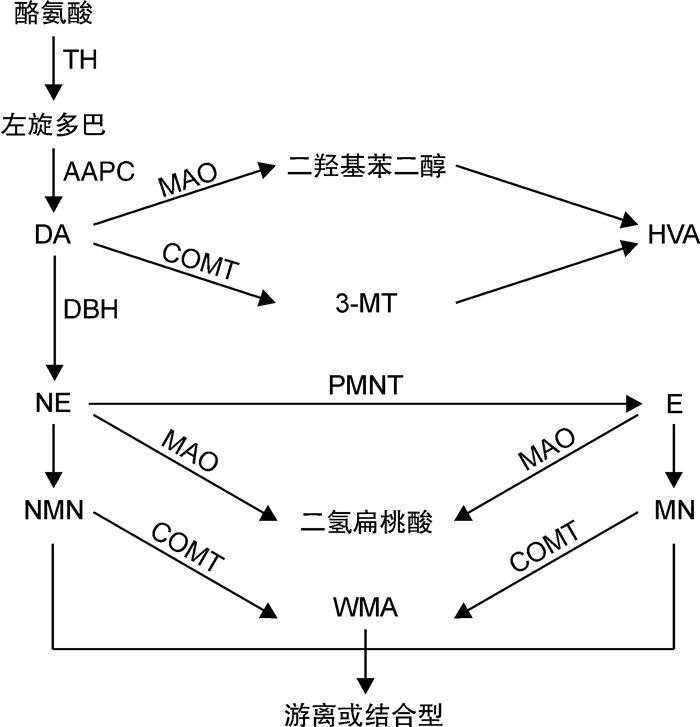
表 1 不同体液MNs检测特点及影响因素
生化检测 敏感性/% 特异性/% 优点 缺点* 受影响因素 血浆MNs[11] 90~100 79~98 仰卧位最准确(首选) 有创 假阳性:包括生理变异、药物干扰和检测技术异常。测定血尿MNs的假阳性率均为19%~21%[3]。药物或食物如三环类抗抑郁药、拟交感神经药物及含高浓度生物胺的坚果等影响CA分泌,患严重疾病者也可出现假阳性,可通过可乐定抑制试验排除假阳性。受年龄影响的只有血浆游离NMN,5~17岁患者为0.47 nmol/L,随年龄增长正常参考值上限均有不同幅度增加,至60岁以上为1.05 nmol/L[12]。 夜尿MNs[13] 93.7 94.3 无创,方便# 收集不规范(调整肌酐值使MNs正常化可显示较高准确性) 24h尿MNs[13] 87.3 94.2 无创,方便# 日间交感活动及饮食影响较大 唾液MNs[14] 89.0 87.0 无创,方便# 与血浆MNs相关性强 3-MT[15] 97.2~98.6 可识别肾上腺外、SDHx突变和HNPGLs及只产生DA的PPGL - *生化检测结果易受多因素影响:包括年龄、性别、饮食、药物、取样方法和患者姿势等,采血前禁食一夜并仰卧位休息20~30 min可降低假阳性。#对无法血浆检测或达不到仰卧休息20 min后采血的中心最大限度减少不便。 表 2 不同功能成像的适用范围
目标受体 示踪物 首选 次选 当18F-FDOPA或86Ga-DOTA不可用 NET 123I-MIBG ·散发性PPGL(敏感性和特异性为83%~100%和95%~100%)[6] ·转移性PPGL放射治疗前评估 ·遗传性PPGL(除SDHx外) ·转移性、肾上腺外、多病灶及SDHx突变 LAT 18F-FDOPA ·遗传性PPGL如NF1、RET、VHL、HIF2A突变(除SDHx外)[25] 肾上腺外、多病灶、转移性和SDHD相关或原发HNPGL - SSTR 68Ga-DOTA (首选) ·一般PPGL ·多病灶、转移性、SDHD相关或原发HNPGL[28-29] ·SDHx突变[30] ·肾上腺外(因PGL主要过表达SSTR2,68Ga对SSTR2亲和力最高)[31] ·散发性或遗传性PPGL - GLUT 18F-FDG - ·肾上腺外、多病灶、转移性PPGL和SDHx突变 ·散发性PPGL或遗传性PPGL ·转移性、肾上腺外、多病灶 -
[1] Ryder SJ, Love AJ, Duncan EL, et al. PET detectives: Molecular imaging for phaeochromocytomas and paragangliomas in the genomics era[J]. Clin Endocrinol(Oxf), 2021, 95(1): 13-28. doi: 10.1111/cen.14375
[2] Lenders J, Kerstens MN, Amar L, et al. Genetics, diagnosis, management and future directions of research of phaeochromocytoma and paraganglioma: a position statement and consensus of the Working Group on Endocrine Hypertension of the European Society of Hypertension[J]. J Hypertens, 2020, 38(8): 1443-1456. doi: 10.1097/HJH.0000000000002438
[3] Patel D, Phay JE, Yen T, et al. Update on pheochromocytoma and paraganglioma from the SSO Endocrine/Head and neck disease-site work group. part 1 of 2: advances in pathogenesis and diagnosis of pheochromocytoma and paraganglioma[J]. Ann Surg Oncol, 2020, 27(5): 1329-1337. doi: 10.1245/s10434-020-08220-3
[4] Kebebew E. Adrenal incidentaloma[J]. N Engl J Med, 2021, 384(16): 1542-1551. doi: 10.1056/NEJMcp2031112
[5] De Filpo G, Maggi M, Mannelli M, et al. Management and outcome of metastatic pheochromocytomas/paragangliomas: an overview[J]. J Endocrinol Invest, 2021, 44(1): 15-25. doi: 10.1007/s40618-020-01344-z
[6] Garcia-Carbonero R, Matute Teresa F, Mercader-Cidoncha E, et al. Multidisciplinary practice guidelines for the diagnosis, genetic counseling and treatment of pheochromocytomas and paragangliomas[J]. Clin Transl Oncol, 2021, 23(10): 1995-2019. doi: 10.1007/s12094-021-02622-9
[7] 邓建华, 李汉忠. 遗传相关的嗜铬细胞瘤/副神经节瘤的研究进展[J]. 临床泌尿外科杂志, 2013, 28(1): 72-77. doi: 10.13201/j.issn.1001-1420.2013.01.028
[8] La Batide-Alanore A, Chatellier G, Plouin PF. Diabetes as a marker of pheochromocytoma in hypertensive patients[J]. J Hypertens, 2003, 21(9): 1703-1707. doi: 10.1097/00004872-200309000-00020
[9] 中华医学会内分泌学分会. 嗜铬细胞瘤和副神经节瘤诊断治疗专家共识(2020版)[J]. 中华内分泌代谢杂志, 2020, 36(9): 737-750. doi: 10.3760/cma.j.cn311282-20200629-00482
[10] Ku EJ, Kim KJ, Kim JH, et al. Diagnosis for Pheochromocytoma and Paraganglioma: A Joint Position Statement of the Korean Pheochromocytoma and Paraganglioma Task Force[J]. Endocrinol Metab(Seoul), 2021, 36(2): 322-338. doi: 10.3803/EnM.2020.908
[11] Eijkelenkamp K, van Geel EH, Kerstens MN, et al. Blood sampling for metanephrines comparing venipuncture vs. indwelling intravenous cannula in healthy subjects[J]. Clin Chem Lab Med, 2020, 58(10): 1681-1686. doi: 10.1515/cclm-2020-0022
[12] Lenders J, Eisenhofer G. Update on modern management of pheochromocytoma and paraganglioma[J]. Endocrinol Metab(Seoul), 2017, 32(2): 152-161. doi: 10.3803/EnM.2017.32.2.152
[13] Peitzsch M, Kaden D, Pamporaki C, et al. Overnight/first-morning urine free metanephrines and methoxytyramine for diagnosis of pheochromocytoma and paraganglioma: is this an option?[J]. Eur J Endocrinol, 2020, 182(5): 499-509. doi: 10.1530/EJE-19-1016
[14] Eijkelenkamp K, Osinga TE, van Faassen M, et al. Diagnostic accuracy of salivary metanephrines in pheochromocytomas and paragangliomas[J]. Clin Chem, 2021, 67(8): 1090-1097. doi: 10.1093/clinchem/hvab064
[15] Liu L, Xie W, Song Z, et al. Addition of 3-methoxytyramine or chromogranin A to plasma free metanephrines as the initial test for pheochromocytoma and paraganglioma: Which is the best diagnostic strategy[J]. Clin Endocrinol(Oxf), 2022, 96(2): 132-138. doi: 10.1111/cen.14585
[16] Tsoli M, Daskalakis K, Kassi E, et al. A critical appraisal of contemporary and novel biomarkers in pheochromocytomas and adrenocortical tumors[J]. Biology, 2021, 10(7): 580. doi: 10.3390/biology10070580
[17] Osinga TE, Kema IP, Kerstens MN, et al. No influence of antihypertensive agents on plasma free metanephrines[J]. Clin Biochem, 2016, 49(18): 1368-1371. doi: 10.1016/j.clinbiochem.2016.06.004
[18] Richard VR, Zahedi RP, Eintracht S, et al. An LC-MRM assay for the quantification of metanephrines from dried blood spots for the diagnosis of pheochromocytomas and paragangliomas[J]. Anal Chim Acta, 2020, 1128: 140-148. doi: 10.1016/j.aca.2020.06.020
[19] Jian M, Huang H, Li K, et al. A 3-min UPLC-MS/MS method for the simultaneous determination of plasma catecholamines and their metabolites: Methodsverification and diagnostic efficiency[J]. Clin Biochem, 2021, 87: 67-73. doi: 10.1016/j.clinbiochem.2020.10.009
[20] Bílek R, Vlček P, Šafařík L, et al. Chromogranin A in the laboratory diagnosis of pheochromocytoma and paraganglioma[J]. Cancers(Basel), 2019, 11(4): 111.
[21] Parisien-La Salle S, Provençal M, Bourdeau I. Chromogranin A in a Cohort of Pheochromocytomas and Paragangliomas: Usefulness at Diagnosis and as an Early Biomarker of Recurrence[J]. Endocr Pract, 2021, 27(4): 318-325. doi: 10.1016/j.eprac.2020.09.011
[22] Moog S, Castinetti F, Docao C, et al. Recurrence-free survival analysis in locally advanced pheochromocytoma: first appraisal[J]. J Clin Endocrinol Metab, 2021, 106(9): 2726-2737. doi: 10.1210/clinem/dgab202
[23] Li H, Abbas KS, Abdelazeem B, et al. A predictive nomogram for early death in pheochromocytoma and paraganglioma[J]. Front Oncol, 2022, 12: 770958. doi: 10.3389/fonc.2022.770958
[24] Sbardella E, Grossman AB. Pheochromocytoma: An approach to diagnosis[J]. Best Pract Res Clin Endocrinol Metab, 2020, 34(2): 101346. doi: 10.1016/j.beem.2019.101346
[25] Taïeb D, Hicks RJ, Hindié E, et al. European Association of Nuclear Medicine Practice Guideline/Society of Nuclear Medicine and Molecular Imaging Procedure Standard 2019 for radionuclide imaging of phaeochromocytoma and paraganglioma[J]. Eur J Nucl Med Mol Imaging, 2019, 46(10): 2112-2137. doi: 10.1007/s00259-019-04398-1
[26] Cavallaro G, Tarallo M, Chiappini A, et al. Surgical management of adrenocortical carcinoma: current highlights[J]. Biomedicines, 2021, 9(8): 114.
[27] De Filpo G, Cantini G, Rastrelli G, et al. Management and outcome of metastatic pheochromocytomas/paragangliomas: a monocentric experience[J]. J Endocrinol Invest, 2022, 45(1): 149-157. doi: 10.1007/s40618-021-01629-x
[28] Mercado-Asis LB, Wolf KI, Jochmanova I, et al. Pheochromocytoma: A genetic and diagnostic update[J]. Endocrine Practice, 2018, 24(1): 78-90. doi: 10.4158/EP-2017-0057
[29] Janssen I, Chen CC, Taieb D, et al. 68Ga-DOTATATE PET/CT in the localization of head and neck paragangliomas compared with other functional imaging modalities and CT/MRI[J]. J Nucl Med, 2016, 57(2): 186-191. doi: 10.2967/jnumed.115.161018
[30] Taieb D, Jha A, Treglia G, et al. Molecular imaging and radionuclide therapy of pheochromocytoma and paraganglioma in the era of genomic characterization of disease subgroups[J]. Endocr Relat Cancer, 2019, 26(11): R627-R652. doi: 10.1530/ERC-19-0165
[31] Han S, Suh CH, Woo S, et al. Performance of 68Ga-DOTA-Conjugated Somatostatin Receptor-Targeting Peptide PET in Detection of Pheochromocytoma and Paraganglioma: A Systematic Review and Metaanalysis[J]. J Nucl Med, 2019, 60(3): 369-376. doi: 10.2967/jnumed.118.211706
[32] Liu X, Zhang X, Tang J, et al. Dual-targeting nanoprobe for early diagnosis of pheochromocytoma though coinstantaneous identification of circulating tumor cells[J]. Anal Chem, 2021, 93(26): 9036-9040. doi: 10.1021/acs.analchem.1c01977
[33] Udager AM, Magers MJ, Goerke DM, et al. The utility of SDHB and FH immunohistochemistry in patients evaluated for hereditary paraganglioma-pheochromocytoma syndromes[J]. Hum Pathol, 2018, 71: 47-54. doi: 10.1016/j.humpath.2017.10.013
[34] 林登强, 祝宇. 嗜铬细胞瘤的诊断进展[J]. 国际泌尿系统杂志, 2016, 36(5): 782-788. https://www.cnki.com.cn/Article/CJFDTOTAL-PXDM201903017.htm
[35] Lloyd RV. Adrenal cortical tumors, pheochromocytomas and paragangliomas[J]. Mod Pathol, 2011, 24 Suppl 2: S58-65.
[36] 徐云泽, 祝宇, 陈东宁, 等. 联合信号转导和转录激活因子3与热休克蛋白90鉴别良恶性嗜铬细胞瘤的探讨[J]. 临床泌尿外科杂志, 2014, 29(3): 222-224, 227. doi: 10.13201/j.issn.1001-1420.2014.03.012
[37] Farhat NA, Powers JF, Shepard-Barry A, et al. A previously unrecognized monocytic component of pheochromocytoma and paraganglioma[J]. Endocr Pathol, 2019, 30(2): 90-95. doi: 10.1007/s12022-019-9575-6
[38] Niu D, Bai Y, Yao Q, et al. AQP2 as a diagnostic immunohistochemical marker for pheochromocytoma and/or paraganglioma[J]. Gland Surg, 2020, 9(2): 200-208. doi: 10.21037/gs.2020.01.19
[39] Antonio K, Valdez M, Mercado-Asis L, et al. Pheochromocytoma/paraganglioma: recent updates in genetics, biochemistry, immunohistochemistry, metabolomics, imaging and therapeutic options[J]. Gland Surg, 2020, 9(1): 105-123. doi: 10.21037/gs.2019.10.25
[40] Thompson L, Gill AJ, Asa SL, et al. Data set for the reporting of pheochromocytoma and paraganglioma: explanations and recommendations of the guidelines from the International Collaboration on Cancer Reporting[J]. Hum Pathol, 2021, 110: 83-97. doi: 10.1016/j.humpath.2020.04.012
[41] Juhlin CC. Challenges in paragangliomas and pheochromocytomas: from histology to molecular immunohistochemistry[J]. Endocr Pathol, 2021, 32(2): 228-244. doi: 10.1007/s12022-021-09675-0
[42] Papathomas TG, Suurd D, Pacak K, et al. What have we learned from molecular biology of paragangliomas and pheochromocytomas?[J]. Endocr Pathol, 2021, 32(1): 134-153.
[43] Lalli E, Luconi M. The next step: mechanisms driving adrenocortical carcinoma metastasis[J]. Endocr Relat Cancer, 2018, 25(2): R31-R48. doi: 10.1530/ERC-17-0440
[44] Calsina B, Castro-Vega LJ, Torres-Pérez R, et al. Integrative multi-omics analysis identifies a prognostic miRNA signature and a targetable miR-21-3p/TSC2/mTOR axis in metastatic pheochromocytoma/paraganglioma[J]. Theranostics, 2019, 9(17): 4946-4958.
[45] Richter S, Gieldon L, Pang Y, et al. Metabolome-guided genomics to identify pathogenic variants in isocitrate dehydrogenase, fumarate hydratase, and succinate dehydrogenase genes in pheochromocytoma and paraganglioma[J]. Genet Med, 2019, 21(3): 705-717.
[46] Bouça B, Bogalho P, Rizzo M, et al. The role of the metabolome and non-coding RNA on pheochromocytomas and paragangliomas: an update[J]. Metabolites, 2022, 12(2): 111.
[47] Kim HM, Koo JS. Expression of glutamine metabolism-related and amino acid transporter proteins in adrenal cortical neoplasms and pheochromocytomas[J]. Dis Markers, 2021, 2021: 8850990.
-

| 引用本文: | 侯倩, 张彪, 罗瑶, 等. 嗜铬细胞瘤诊断研究进展[J]. 临床泌尿外科杂志, 2022, 37(12): 946-951. doi: 10.13201/j.issn.1001-1420.2022.12.012 |
| Citation: | HOU Qian, ZHANG Biao, LUO Yao, et al. Progress in diagnosis of pheochromocytoma[J]. J Clin Urol, 2022, 37(12): 946-951. doi: 10.13201/j.issn.1001-1420.2022.12.012 |
- Figure 1.



 下载:
下载: