Repairing effect of apigenin-7-glucoside on oxalate-mediated HK-2 cell damage via the lRE1/ASK1/P38 MAPK pathway
-
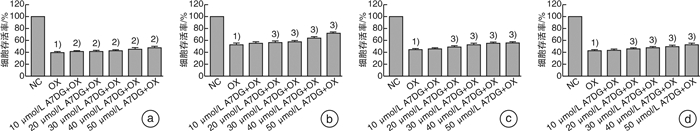
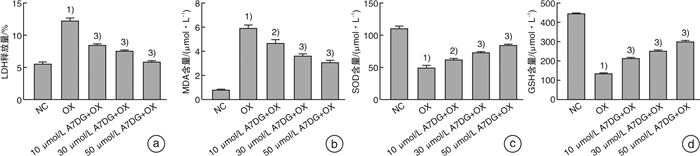
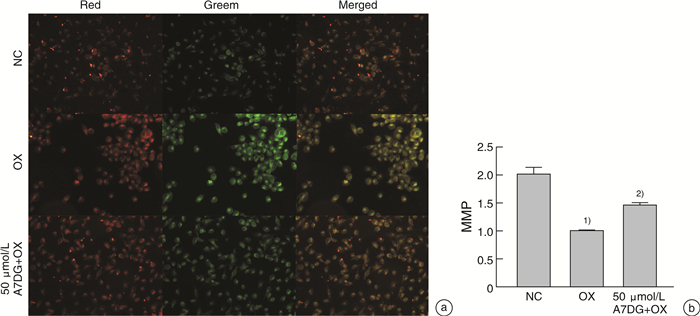
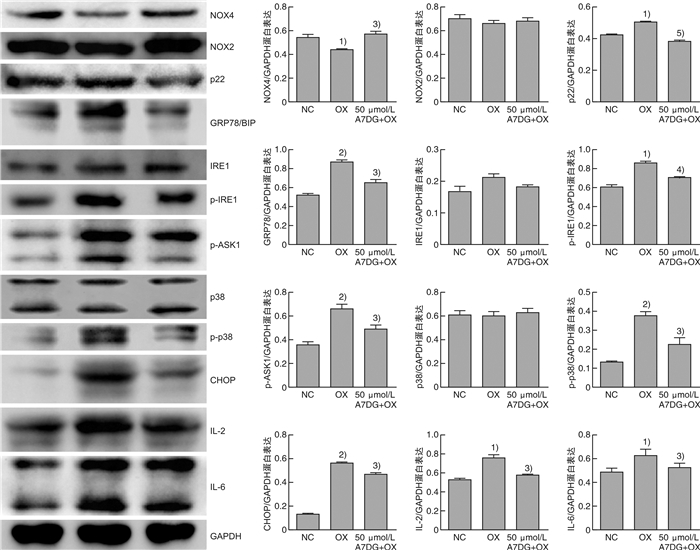
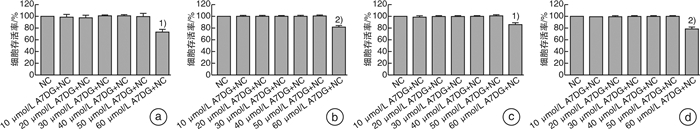
摘要: 目的 探讨自然界中常见的类黄酮糖苷化合物芹菜素-7-葡萄糖苷(A7DG)对草酸诱导的人近端肾小管上皮细胞HK-2损伤是否具有修复作用及其具体作用途径。方法 培养HK-2细胞到80%生长密度后分成3组并进行不同处理:①NC组:换用基础培养液继续培养;②OX组:换用含浓度为2 mmol/L草酸的基础培养液进行培养;③A7DG药物干预组:换用含2 mmol/L草酸和不同浓度的A7DG的基础培养液进行培养。经不同条件处理24 h后,分别对3组HK-2细胞进行细胞活力检测(CCK-8)、乳酸脱氢酶(LDH)及抗氧化能力相关指标还原型谷胱甘肽(GSH)、超氧化物歧化酶(SOD)、丙二醛(MDA)检测,使用荧光显微镜检测线粒体膜电位(MMP)的变化,使用蛋白质印迹法(Western blot)检测细胞内NOX4、NOX2、p22、GRP78/BIP、IRE1、p-IRE1、p-ASK1、p38、p-p38、CHOP以及炎症相关因子IL-2、IL-6的蛋白表达情况。结果 CCK-8实验结果表明OX组细胞存活率较NC组明显降低,而A7DG减轻了OX对HK-2细胞的损伤。LDH、GSH、SOD及MDA检测结果表明A7DG可降低草酸预处理后HK-2细胞NADPH氧化酶的释放,通过干预草酸介导的活性氧(ROS)减轻草酸对HK-2细胞产生的损伤,并使异常的MMP恢复至正常。此外,蛋白质印迹显示A7DG干预下氧化应激特征蛋白如GRP78,CHOP以及IRE1途径关键蛋白p-IRE1、p-ASK、p-MAPK的蛋白表达显著降低,氧化应激损伤途径中IL-2、IL-6等炎性因子的表达也降低。结论 本研究结果证实A7DG在草酸诱导的HK-2细胞损伤中具有积极修复作用,且通过IRE1/ASK1/P38MAPK信号通路发挥积极地抗氧化作用,A7DG可为草酸介导的肾损伤和尿路结石的治疗提供新的参考。Abstract: Objective To investigate whether apigenin-7-glucoside (A7DG) has a repairing effect on oxalic acid-induced damage in HK-2 cells and its specific mechanism.Methods HK-2 cells were cultured to 80% growth density and divided into 3 groups for different treatments: ①NC group: cultured in basal medium; ②OX group: cultured in basal medium containing 2 mmol/L oxalic acid concentration; ③A7DG drug intervention group: cultured in basal medium containing 2 mmol/L oxalic acid and different concentrations of A7DG. After 24 hour of treatment, three groups of HK-2 cells were extracted for cell viability test (CCK-8), lactate dehydrogenase (LDH) test and related antioxidant capacity-related indicators such as reduced glutathione (GSH), superoxide dismutase (SOD), malondialdehyde (MDA) test. Mitochondrial membrane potential (MMP), the protein expression of NOX4, NOX2, p22, GRP78/BIP, IRE1, p-IRE1, p-ASK1, p38, p-p38, CHOP and inflammation-related factors IL-2 and IL-6 were also tested.Results The results of CCK-8 assay showed that the cell growth rate of OX group was significantly lower than that of NC group, and A7DG attenuated the damage of OX on HK-2 cells. The results of LDH, GSH, SOD and MDA assays showed that A7DG reduced the release of NADPH oxidase from HK-2 cells after oxalic acid pretreatment, attenuated the damage of oxalic acid on HK-2 cells by interfering with oxalic acid-mediated reactive oxygen species (ROS) damage and restored the abnormal mitochondrial membrane potential to normal. In addition, the results of WB test showed that the protein expression of oxidative stress signature proteins such as GRP78, CHOP and key proteins of the IRE1 pathway, p-IRE1, p-ASK and p-MAPK, as well as the expression of inflammatory factors such as IL-2 and IL-6 in the oxidative stress damage pathway were significantly reduced by A7DG intervention.Conclusion The results of this study confirmed that A7DG has a positive repairing effect on oxalic acid-induced HK-2 cell injury and exerts a positive antioxidant effect through the IRE1/ASK1/P38MAPK signaling pathway, which means it may provide a new reference for the treatment of oxalate-mediated renal injury and nephrolithiasis.
-
Key words:
- apigenin-7-glucoside /
- oxalate /
- apoptosis /
- kidney stones /
- oxidative stress /
- reactive oxygen species
-

-
[1] 杨嗣星, 叶章群. 再谈尿路结石治疗中需重视的几个关键问题[J]. 中华泌尿外科杂志, 2018, 39(9): 644-646. doi: 10.3760/cma.j.issn.1000-6702.2018.09.002
[2] Yang SX, Song C, Xiong YH. Current perspectives on urolithiasis management in China[J]. World J Urol, 2020, 38(11): 2997-2998. doi: 10.1007/s00345-019-03026-9
[3] 杨嗣星, 廖文彪, 宋超, 等. 湖北省尿路结石患者的临床流行病学调查[J]. 中华泌尿外科杂志, 2018, 39(9): 647-650. doi: 10.3760/cma.j.issn.1000-6702.2018.09.003
[4] Daudon M, Bazin D, Letavernier E. Randall's plaque as the origin of calcium oxalate kidney stones[J]. Urolithiasis, 2015, 43 Suppl 1: 5-11.
[5] Khan SR, Canales BK, Dominguez-Gutierrez PR. Randall's plaque and calcium oxalate stone formation: role for immunity and inflammation[J]. Nat Rev Nephrol, 2021, 17(6): 417-433. doi: 10.1038/s41581-020-00392-1
[6] Rahman N, Khan H, Zia A, et al. Bcl-2 Modulation in p53 Signaling Pathway by Flavonoids: A Potential Strategy towards the Treatment of Cancer[J]. Int J Mol Sci, 2021, 22(21): 11315. doi: 10.3390/ijms222111315
[7] Fidelis QC, Faraone I, Russo D, et al. Chemical and Biological insights of Ouratea hexasperma(A. St. -Hil. )Baill. : a source of bioactive compounds with multifunctional properties[J]. Nat Prod Res, 2019, 33(10): 1500-1503. doi: 10.1080/14786419.2017.1419227
[8] Bian M, Zhang Y, Du X, et al. Apigenin-7-diglucuronide protects retinas against bright light-induced photoreceptor degeneration through the inhibition of retinal oxidative stress and inflammation[J]. Brain Res, 2017, 1663: 141-150. doi: 10.1016/j.brainres.2017.03.019
[9] Nasr Bouzaiene N, Chaabane F, Sassi A, et al. Effect of apigenin-7-glucoside, genkwanin and naringenin on tyrosinase activity and melanin synthesis in B16F10 melanoma cells[J]. Life Sci, 2016, 144: 80-85. doi: 10.1016/j.lfs.2015.11.030
[10] Kim MA, Kang K, Lee HJ, et al. Apigenin isolated from Daphne genkwa Siebold et Zucc. inhibits 3T3-L1 preadipocyte differentiation through a modulation of mitotic clonal expansion[J]. Life Sci, 2014, 101(1-2): 64-72. doi: 10.1016/j.lfs.2014.02.012
[11] Nakazaki E, Tsolmon S, Han J, et al. Proteomic study of granulocytic differentiation induced by apigenin 7-glucoside in human promyelocytic leukemia HL-60 cells[J]. Eur J Nutr, 2013, 52(1): 25-35. doi: 10.1007/s00394-011-0282-4
[12] Samet I, Villareal MO, Motojima H, et al. Olive leaf components apigenin 7-glucoside and luteolin 7-glucoside direct human hematopoietic stem cell differentiation towards erythroid lineage[J]. Differentiation, 2015, 89(5): 146-155. doi: 10.1016/j.diff.2015.07.001
[13] Kumar D, Bhat ZA. Apigenin 7-glucoside from Stachys tibetica Vatke and its anxiolytic effect in rats[J]. Phytomedicine, 2014, 21(7): 1010-1014. doi: 10.1016/j.phymed.2013.12.001
[14] Casas AI, Dao VT, Daiber A, et al. Reactive Oxygen-Related Diseases: Therapeutic Targets and Emerging Clinical Indications[J]. Antioxid Redox Signal, 2015, 23(14): 1171-1185. doi: 10.1089/ars.2015.6433
[15] 赵婷婷, 姜胜, 杨一波, 等. 芹菜素对大鼠心肌缺血再灌注损伤的影响[J]. 中国临床药理学杂志, 2021, 37(20): 2809-2812. https://www.cnki.com.cn/Article/CJFDTOTAL-GLYZ202120023.htm
[16] Alam W, Rocca C, Khan H, et al. Current Status and Future Perspectives on Therapeutic Potential of Apigenin: Focus on Metabolic-Syndrome-Dependent Organ Dysfunction[J]. Antioxidants(Basel), 2021, 10(10): 1643.
[17] Ogboo BC, Grabovyy UV, Maini A, et al. Architecture of the NADPH oxidase family of enzymes[J]. Redox Biol, 2022, 52: 102298. doi: 10.1016/j.redox.2022.102298
[18] Sedeek M, Nasrallah R, Touyz RM, et al. NADPH oxidases, reactive oxygen species, and the kidney: friend and foe[J]. J Am Soc Nephrol, 2013, 24(10): 1512-1518. doi: 10.1681/ASN.2012111112
[19] Liang Y, Liang L, Liu Z, et al. Inhibition of IRE1/JNK pathway in HK-2 cells subjected to hypoxia-reoxygenation attenuates mesangial cells-derived extracellular matrix production[J]. J Cell Mol Med, 2020, 24(22): 13408-13420. doi: 10.1111/jcmm.15964
[20] Bak MJ, Truong VL, Ko SY, et al. Induction of Nrf2/ARE-mediated cytoprotective genes by red ginseng oil through ASK1-MKK4/7-JNK and p38 MAPK signaling pathways in HepG2 cells[J]. J Ginseng Res, 2016, 40(4): 423-430. doi: 10.1016/j.jgr.2016.07.003
[21] Nayak TK, Mamidi P, Sahoo SS, et al. P38 and JNK Mitogen-Activated Protein Kinases Interact With Chikungunya Virus Non-structural Protein-2 and Regulate TNF Induction During Viral Infection in Macrophages[J]. Front Immunol, 2019, 10: 786. doi: 10.3389/fimmu.2019.00786
[22] García-Hernández L, García-Ortega MB, Ruiz-Alcalá G, et al. The p38 MAPK Components and Modulators as Biomarkers and Molecular Targets in Cancer[J]. Int J Mol Sci, 2021, 23(1): 370. doi: 10.3390/ijms23010370
-




 下载:
下载: