Clinical features and prognostic value of TP53 gene mutation in patients with prostate cancer
-
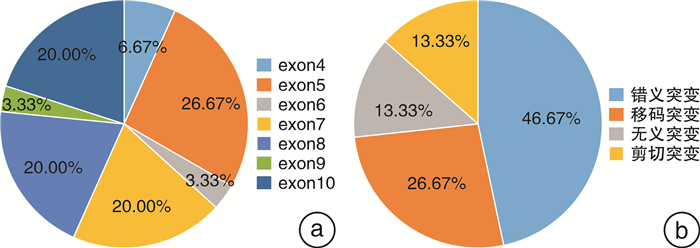
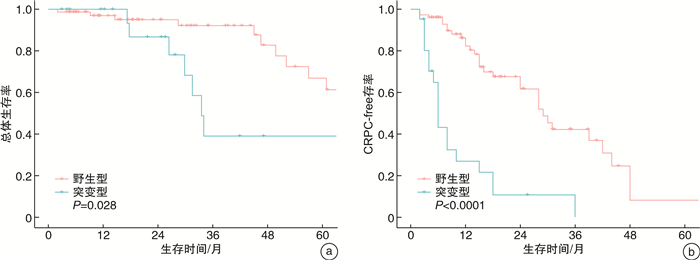
摘要: 目的 探索TP53基因在前列腺癌中的突变图谱,以及TP53突变与患者临床病理特征及预后之间的关系。方法 回顾性收集2011年8月—2022年2月在南京医科大学附属肿瘤医院泌尿外科诊治的97例前列腺癌患者的基本资料、二代测序(NGS)结果及长期随访资料。对比分析TP53突变型和野生型患者临床病理特征和生存结果。通过单因素和多因素Cox回归模型分析TP53突变对总体生存和至去势抵抗性前列腺癌(CRPC)时间的影响。结果 本研究中,TP53突变率为21.65%(21/97);最易发生突变的外显子是第5外显子(26.67%,8/30),最常见的突变类型是错义突变(46.67%,14/30);且组织检测突变的敏感性显著高于外周血。与TP53野生型相比,TP53突变型患者有更年轻的初诊年龄和更高的临床T分期、N分期和M分期。生存分析显示,TP53突变型患者的总体生存和至CRPC时间更短。TP53突变是影响至CRPC时间的独立危险因素。结论 在前列腺癌中,TP53突变与较早的初诊年龄、不良病理特征和较差预后显著相关。此外,TP53突变是影响至CRPC时间的独立危险因素。未来仍需要前瞻性、多中心大样本研究来验证本研究的结果。Abstract: Objective To explore the mutation map of TP53 gene in prostate cancer, and the relationship between TP53 mutation and clinicopathological characteristics as well as prognosis of prostate cancer patients.Methods The basic characteristics, next-generation sequencing (NGS) results and long-term follow-up of 97 prostate cancer patients who were diagnosed and treated in the department of urology, Affiliated Cancer Hospital of Nanjing Medical University from August 2011 to February 2022 were retrospectively collected. The clinicopathological features and survival outcomes of TP53 mutant and wild-type patients were compared. The effects of TP53 mutation on overall survival and time to castration-resistant prostate cancer (CRPC) were analyzed by univariate and multivariate Cox regression analyses.Results In this study, the mutation rate of TP53 was 21.65% (21/97), and exon 5 was the most susceptible to mutation (26.67%, 8/30). The most common mutation type was missense mutation (46.67%, 14/30), and the sensitivity of tissue to detect mutations was significantly higher than that of peripheral blood. Compared with TP53 wild-type, TP53 mutant patients had younger age at diagnosis and higher clinical T, N and M stages. Survival analyses showed that TP53 mutant patients had shorter overall survival and time to CRPC. TP53 mutation was an independent risk factor affecting time to CRPC.Conclusion In prostate cancer, TP53 mutation was significantly associated with early age at diagnosis, poor pathological features, and worse prognosis. In addition, TP53 mutation was an independent risk factor affecting the time to CRPC. Prospective, multi-center and large sample studies are needed to verify the results of this study in the future.
-
Key words:
- prostate cancer /
- TP53 gene /
- prognosis /
- genetic testing /
- mutation
-

-
表 1 入组患者的基本资料及病理特征
M(P25,P75),例(%) 项目 总计(97例) TP53突变型(21例) TP53野生型(76例) P值 年龄/岁 69.00(63.50,74.00) 65.00(58.50,71.00) 70.00(65.00,75.75) 0.027 PSA/(ng·mL-1) 87.24(23.89,290.30) 120.12(47.70,192.50) 73.50(18.875,329.00) 0.342 T分期 0.010 T2 30(30.93) 2(9.52) 28(36.84) T3 43(44.33) 9(42.86) 34(44.74) T4 23(23.71) 10(47.62) 13(17.11) Tx 1(1.03) 0(0.00) 1(1.32) N分期 0.035 N0 36(37.11) 3(14.29) 33(43.42) N1 57(58.76) 17(80.95) 40(52.63) NX 4(4.12) 1(4.76) 3(3.95) M分期 0.001 M0 29(29.90) 0(0.00) 29(38.16) M1 68(70.10) 21(100.00) 47(61.84) ISUP级组 0.518 ISUP 1 1(1.03) 0(0.00) 1(1.32) ISUP 2 11(11.34) 1(4.76) 10(13.16) ISUP 3 10(10.31) 2(9.52) 8(10.53) ISUP 4 27(27.84) 4(19.05) 23(30.26) ISUP 5 48(49.48) 14(66.67) 34(44.74) ISUP:国际泌尿病理学会。 表 2 97例前列腺癌患者总体生存和至CRPC时间多因素分析结果
项目 B SE HR 95%CI P值 总体生存 TP53(突变型vs.野生型) 0.566 0.483 1.761 0.683~4.536 0.241 至CRPC时间 TP53(突变型vs.野生型) 1.421 0.355 4.140 2.066~8.295 < 0.001 CRPC:去势抵抗性前列腺癌。 表 3 7例接受组织和血液配对检测患者的突变情况分析
% 患者序列 血浆特有 组织特有 共有 患者1 0.00 85.71 14.29 患者2 0.00 0.00 100.00 患者3 37.04 48.15 14.81 患者4 3.17 46.03 50.79 患者5 0.00 0.00 100.00 患者6 0.00 100.00 0.00 患者7 0.00 50.00 50.00 平均 5.74 47.13 47.13 -
[1] Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022[J]. CA Cancer J Clin, 2022, 72(1): 7-33. doi: 10.3322/caac.21708
[2] 杨雄, 金泓宇, 宫丽娜, 等. 以多西他赛为基础的新辅助化疗在前列腺癌中的应用进展[J]. 临床泌尿外科杂志, 2021, 38(2): 120-125. doi: 10.13201/j.issn.1001-1420.2021.02.008
[3] Mijit M, Caracciolo V, Melillo A, et al. Role of p53 in the regulation of cellular senescence[J]. Biomolecules, 2020, 10(3): 420. doi: 10.3390/biom10030420
[4] Baugh EH, Ke H, Levine AJ, et al. Why are there hotspot mutations in the TP53 gene in human cancers?[J]. Cell Death Differ, 2018, 25(1): 154-160. doi: 10.1038/cdd.2017.180
[5] Annala M, Vandekerkhove G, Khalaf D, et al. Circulating Tumor DNA genomics correlate with resistance to abiraterone and enzalutamide in prostate cancer[J]. Cancer Discov, 2018, 8(4): 444-457. doi: 10.1158/2159-8290.CD-17-0937
[6] Dong B, Fan L, Yang B, et al. Use of circulating tumor dna for the clinical management of metastatic castration-resistant prostate cancer: a multicenter, real-world study[J]. J Natl Compr Canc Netw, 2021, 19(8): 905-914. doi: 10.6004/jnccn.2020.7663
[7] Ehdaie B, Carlsson S, Vickers A. Racial disparities in low-risk prostate cancer[J]. JAMA, 2019, 321(17): 1726-1727.
[8] Ateeq B, Bhatia V, Goel S. Molecular discriminators of racial disparities in prostate cancer[J]. Trends Cancer, 2016, 2(3): 116-120. doi: 10.1016/j.trecan.2016.01.005
[9] Liu Z, Guo H, Zhu Y, et al. TP53 alterations of hormone-naïve prostate cancer in the Chinese population[J]. Prostate Cancer Prostatic Dis, 2021, 24(2): 482-491. doi: 10.1038/s41391-020-00302-3
[10] 张挺维, 韦煜, 潘剑, 等. TP53突变在中国前列腺癌患者中的临床特征及预后价值研究[J]. 中华外科杂志, 2021, 59(11): 897-901. https://www.cnki.com.cn/Article/CJFDTOTAL-LCMW202212004.htm
[11] Sun J, Zhang K, Cai Z, et al. Identification of critical pathways and hub genes in TP53 mutation prostate cancer by bioinformatics analysis[J]. Biomark Med, 2019, 13(10): 831-840. doi: 10.2217/bmm-2019-0141
[12] Al Zoubi MS, Otoum R, Alorjani MS, et al. TP53, SPOP and PIK3CA genes status in prostate cancer[J]. Asian Pac J Cancer Prev, 2020, 21(11): 3365-3371. doi: 10.31557/APJCP.2020.21.11.3365
[13] Adorno M, Cordenonsi M, Montagner M, et al. A Mutant-p53/Smad complex opposes p63 to empower TGFbeta-induced metastasis[J]. Cell, 2009, 137(1): 87-98. doi: 10.1016/j.cell.2009.01.039
[14] Sauer L, Gitenay D, Vo C, et al. Mutant p53 initiates a feedback loop that involves Egr-1/EGF receptor/ERK in prostate cancer cells[J]. Oncogene, 2010, 29(18): 2628-2637. doi: 10.1038/onc.2010.24
[15] Wang W, Cheng B, Miao L, et al. Mutant p53-R273H gains new function in sustained activation of EGFR signaling via suppressing miR-27a expression[J]. Cell Death Dis, 2013, 4: e574. doi: 10.1038/cddis.2013.97
[16] Hamid AA, Gray KP, Shaw G, et al. Compound genomic alterations of TP53, PTEN, and RB1 Tumor suppressors in localized and metastatic prostate cancer[J]. Eur Urol, 2019, 76(1): 89-97. doi: 10.1016/j.eururo.2018.11.045
[17] De Laere B, Oeyen S, Mayrhofer M, et al. TP53 outperforms other androgen receptor biomarkers to predict abiraterone or enzalutamide outcome in metastatic castration-resistant prostate cancer[J]. Clin Cancer Res, 2019, 25(6): 1766-1773. doi: 10.1158/1078-0432.CCR-18-1943
[18] Kaur HB, Lu J, Guedes LB, et al. TP53 missense mutation is associated with increased tumor-infiltrating T cells in primary prostate cancer[J]. Hum Pathol, 2019, 87: 95-102. doi: 10.1016/j.humpath.2019.02.006
[19] Zou M, Toivanen R, Mitrofanova A, et al. Transdifferentiation as a mechanism of treatment resistance in a mouse model of castration-resistant prostate cancer[J]. Cancer Discov, 2017, 7(7): 736-749. doi: 10.1158/2159-8290.CD-16-1174
-




 下载:
下载: