Establishment of prediction models and modified staging systems for the survival of urachal carcinoma: a real-world study based on SEER database
-
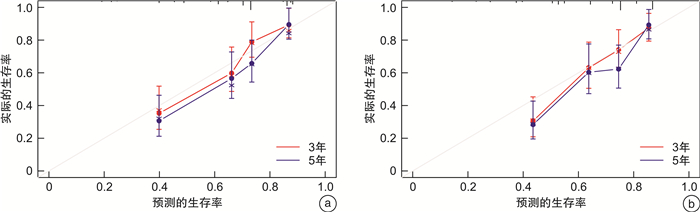
摘要: 目的 基于SEER数据库构建并验证脐尿管癌患者的预后列线图模型并建立改良的分期系统。方法 回顾性检索2010—2018年脐尿管癌患者的临床信息,将患者队列随机以6:4比例分配为训练队列和验证队列。在单因素、多因素Cox回归分析以及双向逐步回归分析的基础上,筛选出独立危险因素并建立列线图。分别采用C指数、受试者工作特征(receiver operating characteristic,ROC)曲线和曲线下面积(area under curve,AUC)值、校准曲线、决策曲线分析(decision curve analysis,DCA)来验证列线图预测准确性。最后使用患者新列线图的评分结合生存信息建立新的分期系统。结果 共筛选出413例患者,分为训练队列247例,验证队列166例。经单因素、多因素Cox回归分析和双向逐步回归分析后,确定年龄、N分期、M分期、Mayo分期及Sheldon分期为脐尿管癌的独立预后因素(P<0.05),其中Mayo分期与Sheldon分期二者呈线性相关,因此分别以二者为基础建立列线图预测模型,C指数分别为0.71±0.02、0.71±0.02。对于3年、5年ROC曲线的AUC值,在以Mayo分期为基础的模型在训练集分别为75.1%,74.9%,在验证集分别为82.2%、72.2%;在以Sheldon分期为基础的模型在训练集分别为75.5%、75.5%,在验证集分别为80.5%、73.3%。校正曲线显示2个模型对患者预后的预测值和实际值一致性较好。DCA曲线显示列线图获益良好。基于列线图总分得到的新风险分期的生存曲线显示新的分期系统分层效果显著。结论 本研究分别基于Mayo分期和Sheldon分期构建了可准确预测脐尿管癌患者预后的列线图模型,并建立了新的分期系统。Abstract: Objective To establish and validate the prognosis nomogram models and modified staging systems of urachal carcinoma patients based on the SEER database.Methods We retrospectively retrieved the clinical information of urachal carcinoma patients from the SEER database diagnosed from 2010 to 2018, then randomly assigned the patients cohort into training cohort and testing cohort in a ratio of 6∶4. The independent risk factors were screened and the nomograms were established based on the univariate, multivariate Cox regression analysis and bidirectional stepwise regression analysis. The C-index, receiver operating characteristic(ROC)curve, area under curve (AUC)value, calibration curve and decision curve analysis(DCA)curve were used to validate the prediction ability of the nomogram models. Lastly we constructed the new staging systems based on the nomogram scores combine with survival information.Results A total of 413 patients were screened, 247 in the training cohort and 166 in the testing cohort. After the univariate, multivariate Cox regression and bidirectional stepwise regression analysis, the age, N stage, M stage, Mayo stage and Sheldon stage were determined as independent risk factors of the prognosis of urachal carcinoma patients (P < 0.05). Mayo stage and Sheldon stage were linearly correlated with each other. Thus, we constructed the prognosis prediction nomogram models based on Mayo stage and Sheldon stage seperately, and the C-index were 0.71±0.02, 0.71±0.02. For the AUC values of 3-year and 5-year ROC curves, the Mayo-based model were 75.1% and 74.9% in the training cohort, 82.2% and 72.2% in the testing cohort; the Sheldon-based model were 75.5% and 75.5% in the training cohort, 80.5% and 73.3% in the testing cohort. The calibration curves showed satisfactory consistency between the predictive value and real value of prognosis. The DCA curves showed the new models benefited well. The survival curves of new staging systems established based on the nomogram points showed significant stratification ability. Conclusion This study established prognosis nomogram models that can accurately predict the prognosis of urachal carcinoma patients based on Mayo and Sheldon staging systems seperately and also established modified staging systems.
-
Key words:
- urachral carcinoma /
- prognosis /
- nomogram /
- SEER database
-

-
表 1 Sheldon分期和Mayo分期
分期 Sheldon分期 Mayo分期 Ⅰ期 肿瘤仅局限于脐尿管黏膜 肿瘤局限于脐尿管和(或)膀胱 Ⅱ期 肿瘤突破脐尿管黏膜层但仍局限于脐尿管内 肿瘤向脐尿管或膀胱肌层外扩展 Ⅲ期 出现局部淋巴结转移 ⅢA期 肿瘤自脐尿管侵及膀胱 ⅢB期 肿瘤侵及腹壁 ⅢC期 肿瘤侵及腹膜 ⅢD期 肿瘤侵及除膀胱以外其他器官 Ⅳ期 出现远处的淋巴结转移或远处器官转移 ⅣA期 淋巴结转移 ⅣB期 远处转移 表 2 纳入患者的训练队列和验证队列的临床特征基线情况
例(%) 变量 训练队列(247例) 验证队列(166例) P值 变量 训练队列(247例) 验证队列(166例) P值 年龄/岁 0.952 T分期 0.450 <65 161(65.2) 103(62.1) T1 45(18.2) 39(23.5) 65~75 55(22.3) 42(25.3) T2 55(22.3) 43(25.9) >75 31(12.5) 21(12.6) T3 109(44.1) 67(40.4) 性别 0.488 T4 38(15.4) 17(10.2) 男 141(57.1) 89(53.6) N分期 0.880 女 106(42.9) 77(46.4) N0 207(83.8) 141(84.9) 种族 0.636 N1 29(11.7) 17(10.2) 白色人种 192(77.7) 127(76.5) N2 11(4.5) 8(4.8) 黑色人种 26(10.5) 16(9.6) M分期 0.247 其他 29(11.7) 23(13.9) M0 213(86.2) 136(81.9) 病理类型 0.517 M1 34(13.8) 30(18.1) 尿路上皮癌 23(9.3) 21(12.6) Sheldon分期 0.341 腺癌 201(81.4) 130(78.3) Ⅰ 43(17.4) 34(20.5) 鳞癌 7(2.8) 5(3.0) Ⅱ 46(18.6) 36(21.7) 其他 16(6.5) 10(6.1) Ⅲ 98(39.7) 57(34.3) 手术 0.381 Ⅳ 60(24.3) 39(23.5) 无 19(7.7) 17(10.2) Mayo分期 0.378 有 228(92.3) 149(89.8) Ⅰ 89(36.0) 70(42.2) 淋巴结清扫 0.786 Ⅱ 98(39.7) 57(34.3) 无 141(57.1) 97(58.4) Ⅲ 18(7.3) 5(3.0) 有 106(42.9) 69(41.6) Ⅳ 42(17.0) 34(20.5) 放疗 0.281 生存情况 0.549 无 227(91.9) 157(94.6) 生存 122(49.4) 87(52.4) 有 20(8.1) 9(5.4) 死亡 125(50.6) 79(47.6) 化疗 0.398 生存时间/月 64.1±3.7 59.5±4.0 0.411 无 183(74.1) 129(77.7) 有 64(25.9) 37(22.3) 表 3 针对脐尿管癌患者预后独立危险因素的单因素Cox回归分析结果
变量 单因素分析 变量 单因素分析 HR 95%CI P值 HR 95%CI P值 年龄/岁 M分期 <65 Reference M0 Reference 65~75 1.259 0.811~1.953 0.305 M1 2.648 1.731~4.052 <0.001 >75 2.166 1.345~3.487 0.001 手术 性别 无 Reference 男 Reference 有 0.504 0.283~0.897 0.019 女 1.105 0.777~1.574 0.578 淋巴结清扫 种族 无 Reference 白色人种 Reference 有 0.950 0.662~1.366 0.785 黑色人种 0.895 0.501~1.600 0.709 放疗 其他 1.090 0.622~1.910 0.763 无 Reference 病理类型 有 1.456 0.834~2.541 0.186 尿路上皮癌 Reference 化疗 腺癌 1.227 0.657~2.290 0.519 无 Reference 鳞癌 2.571 0.890~7.420 0.080 有 1.254 0.852~1.844 0.250 其他 1.724 0.713~4.169 0.226 Sheldon分期 T分期 Ⅰ Reference T1 Reference Ⅱ 1.097 0.514~2.338 0.811 T2 1.647 0.829~3.272 0.154 Ⅲ 2.229 1.230~4.037 0.008 T3 2.800 1.563~5.016 <0.001 Ⅳ 4.481 2.457~8.172 <0.001 T4 4.269 2.240~8.137 <0.001 Mayo分期 N分期 Ⅰ Reference N0 Reference Ⅱ 2.142 1.342~3.419 0.001 N1 2.027 1.260~3.262 0.004 Ⅲ 2.586 1.275~5.243 0.008 N2 5.009 2.561~9.796 <0.001 Ⅳ 5.405 3.257~8.968 <0.001 表 4 针对脐尿管癌患者预后危险因素分别结合Mayo分期和Sheldon分期的多因素Cox回归分析结果
变量 多因素分析(结合Mayo分期) 多因素分析(结合Sheldon分期) HR 95%CI P值 HR 95%CI P值 年龄/岁 <65 Reference Reference 65~75 1.155 0.738~1.806 0.529 1.100 0.701~1.719 0.683 >75 2.639 1.560~4.464 <0.001 2.906 1.737~4.861 <0.001 N分期 N0 Reference Reference N1 0.777 0.320~1.886 0.577 0.589 0.243~1.429 0.242 N2 0.812 0.173~3.797 0.791 1.802 0.685~4.738 0.233 M分期 M0 Reference Reference M1 0.266 0.051~1.382 0.115 0.826 0.365~1.871 0.647 手术 无 Reference Reference 有 0.779 0.396~1.530 0.468 0.852 0.435~1.672 0.642 Sheldon分期 Ⅰ Reference Ⅱ 1.247 0.580~2.679 0.573 Ⅲ 2.616 1.429~4.793 0.002 Ⅳ 7.061 2.263~22.028 <0.001 Mayo分期 Ⅰ Reference Ⅱ 2.358 1.468~3.786 <0.001 Ⅲ 3.885 1.202~12.558 0.023 Ⅳ 19.226 3.311~111.615 <0.001 -
[1] Guerin M, Miran C, Colomba E, et al. Urachal carcinoma: a large retrospective multicentric study from the French Genito-Urinary Tumor Group[J]. Front Oncol, 2023, 13: 1110003. doi: 10.3389/fonc.2023.1110003
[2] Song SH, Lee J, Ko YH, et al. TNM-Based Head-to-Head Comparison of Urachal Carcinoma and Urothelial Bladder Cancer: Stage-Matched Analysis of a Large Multicenter National Cohort[J]. Cancer Res Treat, 2023, 55(4): 1337-1345. doi: 10.4143/crt.2023.417
[3] Yu YD, Ko YH, Kim JW, et al. The Prognosis and Oncological Predictor of Urachal Carcinoma of the Bladder: A Large Scale Multicenter Cohort Study Analyzed 203 Patients With Long Term Follow-Up[J]. Front Oncol, 2021, 11: 683190. doi: 10.3389/fonc.2021.683190
[4] 黄健, 张旭, 魏强, 等. 中国泌尿外科和男科疾病诊断治疗指南[M]. 北京: 科学出版社, 2022: 83.
[5] Sheldon CA, Clayman RV, Gonzalez R, et al. Malignant urachal lesions[J]. J Urol, 1984, 131(1): 1-8. doi: 10.1016/S0022-5347(17)50167-6
[6] Molina JR, Quevedo JF, Furth AF, et al. Predictors of survival from urachal cancer: a Mayo Clinic study of 49 cases[J]. Cancer, 2007, 110(11): 2434-2440. doi: 10.1002/cncr.23070
[7] 罗贞波, 蔡涛浓, 梁海滔, 等. 单中心初发脐尿管癌55例诊治分析[J]. 临床泌尿外科杂志, 2022, 37(11): 857-860. https://lcmw.whuhzzs.com/article/doi/10.13201/j.issn.1001-1420.2022.11.011
[8] Bruins HM, Visser O, Ploeg M, et al. The clinical epidemiology of urachal carcinoma: results of a large, population based study[J]. J Urol, 2012, 188(4): 1102-1107. doi: 10.1016/j.juro.2012.06.020
[9] Ryan PC, Kelly C, Afridi I, et al. Surgical treatment of urachal remnants in an adult population-a single-centre experience[J]. Ir J Med Sci, 2023, 192(6): 3023-3027. doi: 10.1007/s11845-023-03339-0
[10] Gural Z, Yücel S, Oskero lu S, et al. Urachal adenocarcinoma: A case report and review of the literature[J]. J Cancer Res Ther, 2022, 18(1): 291-293. doi: 10.4103/jcrt.JCRT_28_20
[11] Zaleski MP, Chen H, Roy-Chowdhuri S, et al. Distinct Gene Mutations Are Associated With Clinicopathologic Features in Urachal Carcinoma[J]. Am J Clin Pathol, 2022, 158(2): 263-269. doi: 10.1093/ajcp/aqac039
[12] Guerin M, Miran C, Colomba E, et al. Urachal carcinoma: a large retrospective multicentric study from the French Genito-Urinary Tumor Group[J]. Frontiers in oncology, 2023, 13: 1110003. doi: 10.3389/fonc.2023.1110003
[13] Siefker-Radtke A. Urachal adenocarcinoma: a clinician's guide for treatment[J]. Semin Oncol, 2012, 39(5): 619-624. doi: 10.1053/j.seminoncol.2012.08.011
[14] Dursun F, Lim K, Svatek RS, et al. Clinical outcomes and patterns of population-based management of urachal carcinoma of the bladder: An analysis of the National Cancer Database[J]. Cancer Med, 2022, 11(22): 4273-4282. doi: 10.1002/cam4.4786
[15] Szarvas T, Módos O, Niedworok C, et al. Clinical, prognostic, and therapeutic aspects of urachal carcinoma-A comprehensive review with meta-analysis of 1, 010 cases[J]. Urol Oncol, 2016, 34(9): 388-398. doi: 10.1016/j.urolonc.2016.04.012
[16] 袁翔, 王军, 王涛, 等. 脐尿管癌的临床特征及预后相关因素分析[J]. 中华泌尿外科杂志, 2021, 42(2): 110-115. https://cdmd.cnki.com.cn/Article/CDMD-10459-1021079541.htm
[17] Zhang J, WU J. Options for diagnosis and treatment of urachal carcinoma[J]. Asia Pac J Clin Oncol, 2013, 9(2): 117-122. doi: 10.1111/j.1743-7563.2012.01592.x
[18] Taktak S, El-Taji O, Hanchanale V. Modern methods in managing urachal adenocarcinoma[J]. Curr Urol, 2023, 17(3): 188-192. doi: 10.1097/CU9.0000000000000189
[19] Gopalan A, Sharp DS, Fine SW, et al. Urachal carcinoma: a clinicopathologic analysis of 24 cases with outcome correlation[J]. Am J Surg Pathol, 2009, 33(5): 659-668. doi: 10.1097/PAS.0b013e31819aa4ae
[20] Jung HA, Sun JM, Park SH, et al. Treatment Outcome and Relevance of Palliative Chemotherapy in Urachal Cancer[J]. Chemotherapy, 2014, 60(2): 73-80. doi: 10.1159/000368071
[21] Ding L, Xia B, Zhang Y, et al. Web-Based Prediction Models for Overall Survival and Cancer-Specific Survival of Patients With Primary Urachal Carcinoma: A Study Based on SEER Database[J]. Front Public Health, 2022, 10: 870920. doi: 10.3389/fpubh.2022.870920
-





 下载:
下载: