Clinical study on artificial intelligence-based prediction of bladder cancer recurrence using preoperative CT images, blood and biochemical data
-
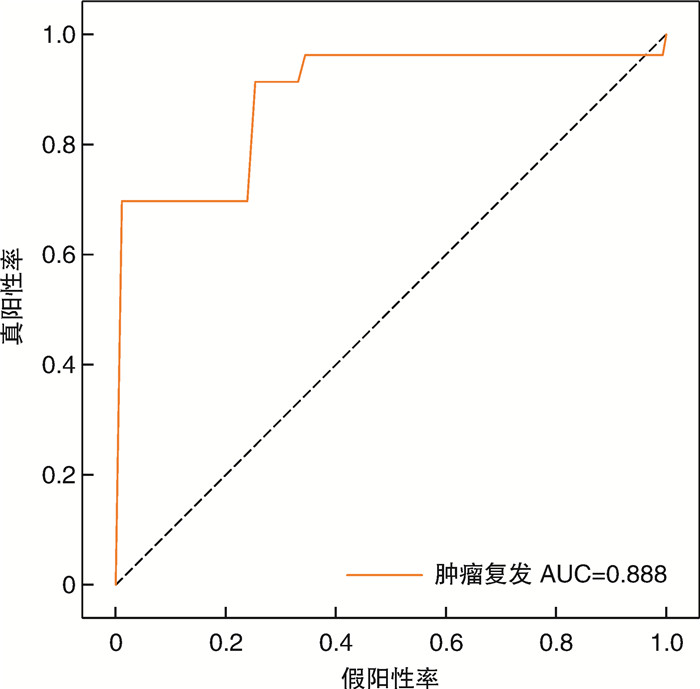
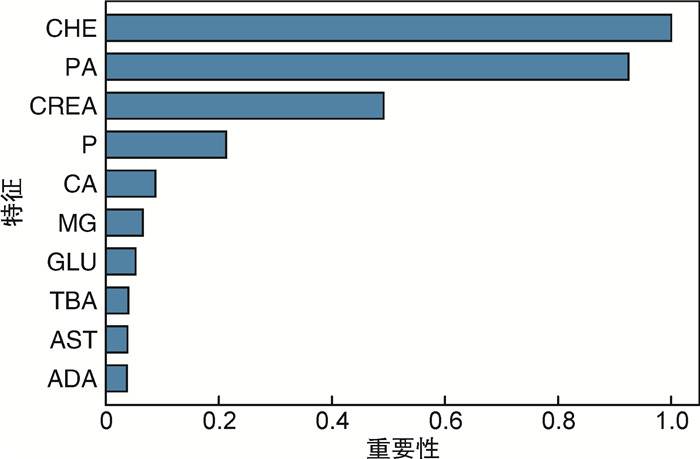
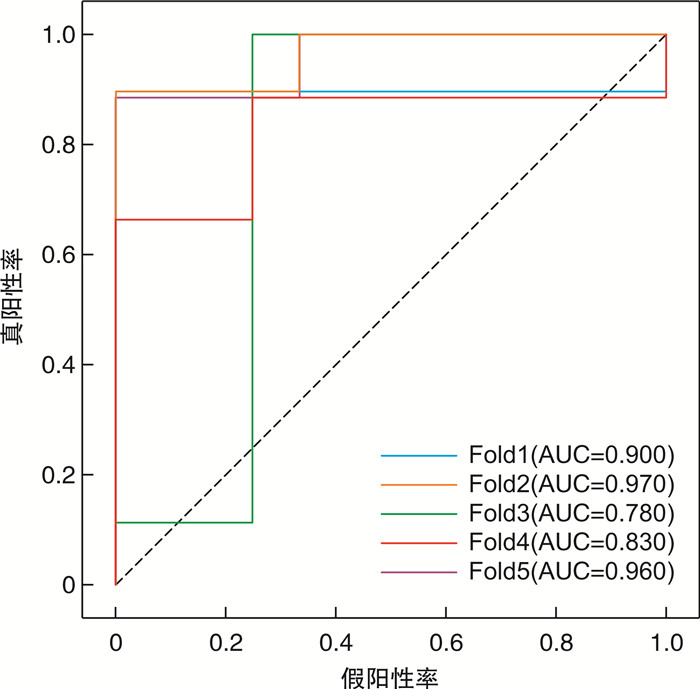
摘要: 目的 本研究旨在探讨CT图像、血常规及生化数据在预测膀胱癌复发风险中的价值。方法 回顾性纳入2017年3月—2022年7月于徐州中心医院泌尿外科治疗的65例膀胱癌患者。当患者初发膀胱癌时,收集其术前CT图像、血常规及生化数据。将CT图像进行归一化,并随机旋转-40~40°,以增加数据输入。使用CT图像、血常规及生化数据分别构建多种预测模型(三维卷积神经网络、梯度提升机)。采用五倍交叉验证实验及曲线下面积(area under the curve,AUC)评价三维卷积神经网络和梯度提升机的预测性能。结果 利用CT图像训练的三维卷积神经网络准确率为89.0%,曲线下面积为0.888。基于血常规和生化数据训练的梯度提升机准确率分别为94.7%、98.8%,曲线下面积分别为0.898和0.996。结论 机器学习方法在对经尿道膀胱肿瘤切除术后患者的复发预测方面显示出巨大的潜力,或可用于膀胱癌的复发风险分层,进一步指导后续的化学治疗。Abstract: Objective To investigate the value of machine learning-based utilization of CT scans, routine blood tests and chemistry panels in predicting the risk of bladder cancer recurrence.Methods Sixty-five patients with bladder cancer treated at the department of urology of Xuzhou Central Hospital in Jiangsu Province were included retrospectively from March 2017 to July 2022. When the patient first developed bladder cancer, the preoperative CT images, blood routine and biochemical data were collected. CT images were normalized, and were also randomly rotated -40 to 40 degrees to increase data input. Two prediction models (3D convolutional neural network and gradient boosting machines) were constructed using CT images, blood routine and biochemical data. Five-fold cross-validation experiments and AUC curve were used to evaluate the prediction performance of 3D convolutional neural network and gradient pusher.Results The accuracy of the 3D convolutional neural network trained by CT images was 89.0%, and the area under the curve was 0.888. The accuracy of the gradient boosting machines trained based on blood routine and biochemical data were 94.7% and 98.8%, and the area under the curve were 0.898 and 0.996, respectively.Conclusion Machine learning approaches show great potential for predicting recurrence in patients after transurethral bladder tumor resection, and may provide insights into the design of risk-level-dependent adjuvant therapies.
-
Key words:
- bladder cancer /
- recurrence /
- artificial intelligence /
- CT images
-

-
表 1 65例患者血常规和生化数据
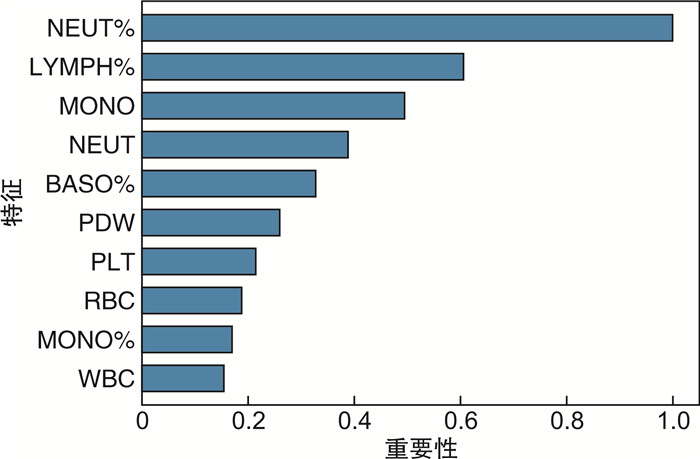
X±S 项目 复发组(17例) 未复发组(48例) 血常规 嗜碱性粒细胞计数/(×109/L) 0.028±0.012 0.032±0.016 嗜酸性粒细胞计数/(×109/L) 0.139±0.148 0.149±0.117 嗜酸性粒细胞百分比/% 2.171±1.728 2.335±1.876 单核细胞计数/(×109/L) 0.489±0.122 0.541±0.225 单核细胞百分比/% 8.029±1.581 8.154±2.285 中性粒细胞计数/(×109/L) 3.881±0.902 4.196±1.876 中性粒细胞百分比/% 63.100±5.981 61.613±11.068 嗜碱性粒细胞百分比/% 0.471±0.145 0.493±0.265 大血小板数/% 28.347±8.671 29.365±8.795 血细胞比容/% 41.988±4.960 40.524±5.821 血红蛋白/(g/L) 138.706±15.695 133.630±20.157 淋巴细胞计数/(×109/L) 1.626±0.506 1.785±0.792 淋巴细胞百分比/% 26.229±5.395 27.404±9.397 平均血红蛋白量/pg 30.700±1.965 30.874±2.110 平均血红蛋白浓度/(g/L) 330.765±11.059 329.565±10.500 红细胞平均体积/fL 92.841±4.912 93.661±5.218 平均血小板体积/fL 10.400±1.077 10.567±1.093 降钙素原/(ng/mL) 0.222±0.053 0.244±0.067 血小板分布宽度/fL 12.859±2.421 12.420±2.250 血小板/(×109/L) 213.118±46.984 234.391±74.012 红细胞计数/(×1012/L) 4.549±0.667 4.345±0.661 白细胞计数/(×109/L) 6.162±1.359 6.703±2.390 生化 谷丙转氨酶/(U/L) 20.714±11.335 20.209±10.746 碱性磷酸酶/(U/L) 94.571±30.246 82.628±18.782 腺苷脱氨酶/(U/L) 9.614±2.529 6.770±2.907 氯/(mmol/L) 101.486±2.505 101.086±2.941 谷草转氨酶/(U/L) 22.000±9.350 20.442±6.135 白蛋白/(g/L) 43.257±3.361 41.160±4.240 钙/(mmol/L) 2.439±0.078 2.344±0.120 直接胆红素/(μmol/L) 3.914±1.843 4.328±2.013 肌酐/(μmol/L) 53.757±10.064 64.523±18.375 葡萄糖/(mmol/L) 5.014±0.418 5.878±1.570 谷氨酰转移酶/(U/L) 28.000±24.779 27.093±18.922 总胆汁酸/(μmol/L) 5.229±3.633 5.509±5.263 尿酸/(μmol/L) 295.757±48.802 302.847±72.048 镁/(mmol/L) 0.894±0.060 0.902±0.136 钠/(mmol/L) 140.200±2.270 140.453±2.581 白蛋白/球蛋白比 1.600±0.245 1.728±0.301 磷/(mmol/L) 1.139±0.109 1.070±0.196 CHE/(U/L) 9 271.714±1 141.615 7 257.047±2 206.267 球蛋白/(g/L) 27.543±4.789 24.472±4.502 总蛋白/(g/L) 70.800±7.332 65.633±7.288 前白蛋白/(mg/L) 286.286±41.039 215.372±63.137 总胆红素/(μmol/L) 11.914±4.039 11.895±5.138 钾/(mmol/L) 4.017±0.297 4.147±0.320 注:CHE:胆碱酯酶。 表 2 CT图像的五倍交叉验证结果
项目 1 2 3 4 5 平均值 准确率 训练集 0.900 0.800 0.820 0.900 0.750 0.834 测试集 0.920 0.920 0.920 0.770 0.920 0.890 AUC 训练集 0.950 0.930 0.930 0.950 0.710 0.894 测试集 0.900 0.970 0.780 0.830 0.960 0.888 表 3 使用血常规的梯度提升机的五倍交叉验证结果
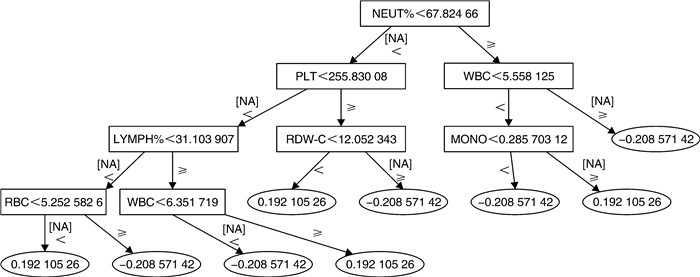
项目 1 2 3 4 5 平均值 训练集 准确率 0.870 1.000 0.870 0.790 1.000 0.906 AUC 0.810 1.000 0.910 0.730 1.000 0.890 测试集 准确率 0.947 AUC 0.898 表 4 使用生化的梯度提升机的五倍交叉验证结果
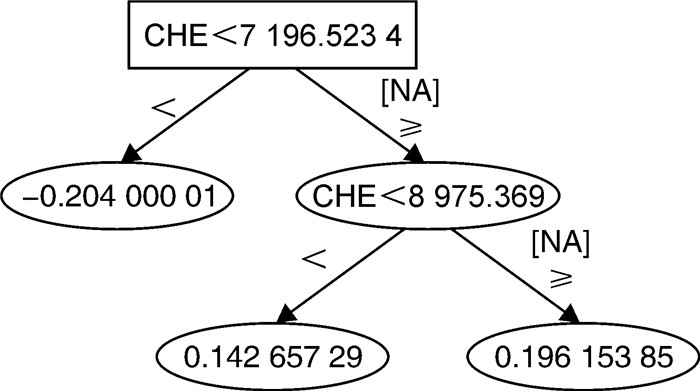
1 2 3 4 5 平均值 训练集 准确率 0.930 1.000 0.930 1.000 1.000 0.972 AUC 0.980 1.000 0.960 1.000 1.000 0.988 测试集 准确率 0.988 AUC 0.996 -
[1] 张瑞赟, 金迪, 钱雷, 等. 肌层浸润性膀胱癌新辅助化疗病理与长期临床结局——单中心15年真实世界临床研究[J]. 临床泌尿外科杂志, 2023, 38(12): 915-920. https://lcmw.whuhzzs.com/article/doi/10.13201/j.issn.1001-1420.2023.12.005
[2] Zhang G, Wu Z, Zhang X, et al. CT-based radiomics to predict muscle invasion in bladder cancer[J]. Eur Radiol, 2022, 32(5): 3260-3268. doi: 10.1007/s00330-021-08426-3
[3] Sylvester RJ, Van Der Meijden AP, Oosterlinck W, et al. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials[J]. Eur Urol, 2006, 49(3): 466-465;discussion 475-477.
[4] Fernandez-Gomez J, Madero R, Solsona E, et al. Predicting nonmuscle invasive bladder cancer recurrence and progression in patients treated with bacillus Calmette-Guerin: the CUETO scoring model[J]. J Urol, 2009, 182(5): 2195-2203. doi: 10.1016/j.juro.2009.07.016
[5] Chang SS, Boorjian SA, Chou R, et al. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline[J]. J Urol, 2016, 196(4): 1021-1029. doi: 10.1016/j.juro.2016.06.049
[6] Kluth LA, Black PC, Bochner BH, et al. Prognostic and Prediction Tools in Bladder Cancer: A Comprehensive Review of the Literature[J]. Eur Urol, 2015, 68(2): 238-253. doi: 10.1016/j.eururo.2015.01.032
[7] Kourou K, Exarchos TP, Exarchos KP, et al. Machine learning applications in cancer prognosis and prediction[J]. Comput Struct Biotechnol J, 2015, 13: 8-17. doi: 10.1016/j.csbj.2014.11.005
[8] Tokuyama N, Saito A, Muraoka R, et al. Prediction of non-muscle invasive bladder cancer recurrence using machine learning of quantitative nuclear features[J]. Mod Pathol, 2022, 35(4): 533-538. doi: 10.1038/s41379-021-00955-y
[9] Hasnain Z, Mason J, Gill K, et al. Machine learning models for predicting post-cystectomy recurrence and survival in bladder cancer patients[J]. PLoS One, 2019, 14(2): e0210976. doi: 10.1371/journal.pone.0210976
[10] Xu Y, Lou J, Gao Z, et al. Computed Tomography Image Features under Deep Learning Algorithm Applied in Staging Diagnosis of Bladder Cancer and Detection on Ceramide Glycosylation[J]. Comput Math MethodsMed, 2022, 2022: 7979523.
[11] Yang Y, Zou X, Wang Y, et al. Application of deep learning as a noninvasive tool to differentiate muscle-invasive bladder cancer and non-muscle-invasive bladder cancer with CT[J]. Eur J Radiol, 2021, 139: 109666. doi: 10.1016/j.ejrad.2021.109666
[12] Cha KH, Hadjiiski L, Chan HP, et al. Bladder Cancer Treatment Response Assessment in CT using Radiomics with Deep-Learning[J]. Sci Rep, 2017, 7(1): 8738. doi: 10.1038/s41598-017-09315-w
[13] Wang DQ, Shuai J, Zheng H, et al. Can Routine Blood and Urine Parameters Reveal Clues to Detect Bladder Cancer? A Case-Control Study[J]. Front Oncol, 2021, 11: 796975.
[14] Zhang J, Zhou X, Ding H, et al. The prognostic value of routine preoperative blood parameters in muscle-invasive bladder cancer[J]. BMC Urol, 2020, 20(1): 31. doi: 10.1186/s12894-020-00602-9
[15] Nakamura M, Kajiwara Y, Otsuka A, et al. LVQ-SMOTE-Learning Vector Quantization based Synthetic Minority Over-sampling Technique for biomedical data[J]. BioData Min, 2013, 6(1): 16. doi: 10.1186/1756-0381-6-16
[16] Zhang G, Wu Z, Xu L, et al. Deep Learning on Enhanced CT Images Can Predict the Muscular Invasiveness of Bladder Cancer[J]. Front Oncol, 2021, 11: 654685. doi: 10.3389/fonc.2021.654685
[17] Qian J, Yang L, Hu S, et al. Feasibility Study on Predicting Recurrence Risk of Bladder Cancer Based on Radiomics Features of Multiphase CT Images[J]. Front Oncol, 2022, 12: 899897. doi: 10.3389/fonc.2022.899897
[18] Chen SY, Du LD, Zhang YH. Pilot study of intravesical instillation of two new generation anthracycline antibiotics in prevention of superficial bladder cancer recurrence[J]. Chin Med J(Engl), 2010, 123(23): 3422-3426.
[19] Lee J, Choo MS, Yoo S, et al. Intravesical Prostatic Protrusion and Prognosis of Non-Muscle Invasive Bladder Cancer: Analysis of Long-Term Data over 5 Years with Machine-Learning Algorithms[J]. J Clin Med, 2021, 10(18): 4263. doi: 10.3390/jcm10184263
[20] D'Andrea D, Moschini M, Gust K, et al. Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Primary Non-muscle-invasive Bladder Cancer[J]. Clin Genitourin Cancer, 2017, 15(5): e755-e764. doi: 10.1016/j.clgc.2017.03.007
[21] Liu K, Zhao K, Wang L, et al. The prognostic values of tumor-infiltrating neutrophils, lymphocytes and neutrophil/lymphocyte rates in bladder urothelial cancer[J]. Pathol Res Pract, 2018, 214(8): 1074-1080. doi: 10.1016/j.prp.2018.05.010
[22] Kimura S, Soria F, D'andrea D, et al. Prognostic Value of Serum Cholinesterase in Non-muscle-invasive Bladder Cancer[J]. Clin Genitourin Cancer, 2018, 16(6): e1123-e1132. doi: 10.1016/j.clgc.2018.07.002
[23] Tang X, Du P, Yang Y. The clinical use of neutrophil-to-lymphocyte ratio in bladder cancer patients: a systematic review and meta-analysis[J]. Int J Clin Oncol, 2017, 22(5): 817-825. doi: 10.1007/s10147-017-1171-5
[24] Huang J, Wang Y, Yuan Y, et al. Preoperative serum pre-albumin as an independent prognostic indicator in patients with localized upper tract urothelial carcinoma after radical nephroureterectomy[J]. Oncotarget, 2017, 30;8(22): 36772-36779.
[25] Qin T, Yu H, Wang B, et al. PD-L1 and neutrophil to lymphocyte ratio(NLR)as predictive panel of prognosis in bladder cancer[J]. Journal of Clinical Oncology, 38(15_suppl): e17040-e17040. doi: 10.1200/JCO.2020.38.15_suppl.e17040
[26] Thomas J, Riaz I B, Freeman D, et al. Early changes in peripheral blood neutrophil-lymphocyte ratio(NLR)to predict outcomes with immune checkpoint inhibitors(ICIs)for metasta tic urothelial carcinoma(mUC)[J]. Journal of Clinical Oncology, 40(6_suppl): 449-449. doi: 10.1200/JCO.2022.40.6_suppl.449
[27] Nair M, Dawsey S, Pramod N, et al. Neutrophil-to-lymphocyte ratio as a predictor of immune checkpoint inh ibitor therapy response in advanced bladder cancer[J]. J Clin Oncol, 41(16_suppl): e16586-e16586. doi: 10.1200/JCO.2023.41.16_suppl.e16586
[28] Sheng IY Diaz-Montero CM, Rayman PA, et al. Blood myeloid derived suppressor cells(MDSC)in metastatic urothelial carcinoma(mUC)are correlated with neutrophil-to-lymphocyte ratio(N LR)and overall survival(OS)[J]. J Clin Oncol, 37(7_suppl): 436-436. doi: 10.1200/JCO.2019.37.7_suppl.436
[29] Wu Y, Zhu W, Wang J, et al. Using machine learning for mortality prediction and risk stratificatio n in atezolizumab-treated cancer patients: Integrative analysis of eig ht clinical trials[J]. Cancer medicine, 12(3): 3744-3757. doi: 10.1002/cam4.5060
-




 下载:
下载: