-
摘要: 目的 探讨儿童肾母细胞瘤临床病理因素与预后的相关性。方法 回顾性分析2008年1月—2020年12月安徽省儿童医院收治的75例肾母细胞瘤患者的临床病理资料,分析患者的年龄、性别、发病部位、临床分期、病理类型、Ki-67增殖指数、治疗方法与预后的相关性。采用SPSS 26.0对数据进行统计分析,Graphpad prism7绘图,Kaplan-Meier生存分析中的log-rank检验对不同组间累积无进展生存率进行比较,Cox比例风险模型筛选出影响患者预后的相关因素。结果 75例患者,男44例、女31例,平均年龄(2.40±2.01)岁。中位随访时间84个月,期间15例死亡。log-rank检验结果显示,年龄、临床分期、病理类型和Ki-67增殖指数与预后有一定的相关性,多变量分析显示,年龄、临床分期和病理类型为影响患者疾病进展或死亡的危险因素。结论 肾母细胞瘤是儿童最常见的泌尿系统肿瘤,早期发现、正确诊断、明确分期能协助临床医生评估个体风险,选择最佳治疗方法,提高生存率,改善预后。Abstract: Objective To investigate the correlations between the clinicopathological factors and prognosis of nephroblastoma in children.Methods The clinicopathological data of 75 children with nephroblastoma treated in Anhui Children's Hospital from January 2008 to December 2020 were analyzed retrospectively. The correlations of prognosis with age, gender, location of onset, clinical stage, pathological type, Ki-67 proliferation index and treatment method were analyzed. The data were statistically analyzed using SPSS 26.0, and relevant charts were drawn with Graphpad Prism 7. In Kaplan-Meier survival analysis, the cumulative progression-free survival rates among various groups were compared using the log-rank test. The relevant factors influencing the prognoses in children were screened using the Cox proportional hazards model.Results A total of 75 children were enrolled, including 44 boys and 31 girls, with an average age of (2.40±2.01) years. During the median follow-up duration of 84 months, 15 children died. On the one hand, the log-rank test showed that age, clinical stage, pathological type and Ki-67 proliferation index were correlated with the prognosis. On the other hand, multivariate analysis demonstrated that age, clinical stage and pathological type were the risk factors influencing disease progression or death.Conclusion Nephroblastoma is the most common tumor of the urinary system in children. Early detection, correct diagnosis and definite staging can help clinicians evaluate individual risk, select the optimal treatment, enhance survival rate and improve prognosis.
-
Key words:
- nephroblastoma /
- children /
- clinicopathological factors /
- prognosis
-

-
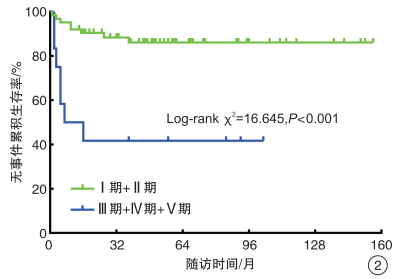
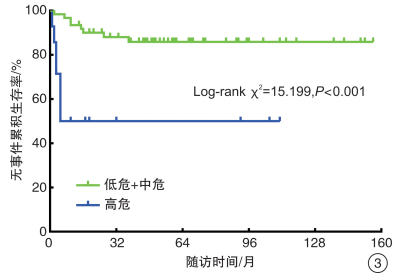
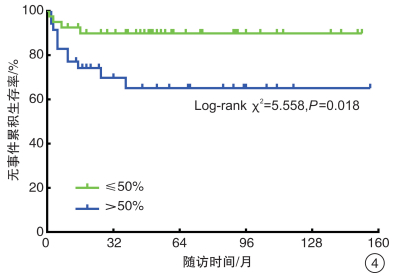
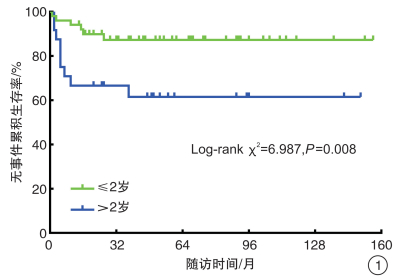
表 1 临床与病理特征和预后的关系
指标 平均生存时间/月 平均生存时间的95%CI log-rank χ2 P值 性别 0.463 0.496 男 117.45 98.66~136.25 女 131.84 112.43~151.25 年龄 6.987 0.008 ≤2岁 137.81 124.14~151.48 >2岁 95.88 67.95~123.81 部位 0.359 0.549 左肾 118.26 101.01~135.52 右肾 121.58 101.79~141.38 临床分期 16.645 < 0.001 Ⅰ期+Ⅱ期 136.42 123.74~149.09 Ⅲ期及以上 46.25 19.04~73.46 病理类型 15.199 < 0.001 低危+中危 136.29 123.58~149 高危 57.21 29.03~85.39 Ki-67 5.558 0.018 ≤50% 137.28 123.6~150.97 >50% 106.20 81.82~130.57 治疗方法 0.009 0.924 先化疗后切除 111.80 86.25~137.35 先切除后化疗 125.11 109.48~140.74 表 2 变量赋值表
变量 赋值 随访时间 数值型 预后 存活=0;转移/死亡=1 年龄 ≤2岁=1;>2岁=2 临床分期 Ⅰ期+Ⅱ期=1;Ⅲ期及以上=2 病理类型 低危+中危=1;高危=2 Ki-67 ≤50%=1;>50%=2 表 3 影响患者预后的Cox比例风险模型
因素 B SE Waldχ2 P HR(95%CI) 年龄 ≤2岁 1.000 >2岁 1.196 0.570 4.409 0.036 3.308(1.083,10.105) 临床分期 Ⅰ期+Ⅱ期 1.000 Ⅲ期及以上 1.532 0.631 5.889 0.015 4.627(1.343,15.944) 病理类型 低危+中危 1.000 高危 1.306 0.649 4.044 0.044 3.690(1.034,13.174) Ki-67 ≤50% 1.000 >50% 1.210 0.631 3.672 0.055 3.353(0.973,11.561) -
[1] Al-Hussain T, Ali A, Akhtar M. Wilms tumor: an update[J]. Adv Anat Pathol, 2014, 21(3): 166-173. doi: 10.1097/PAP.0000000000000017
[2] Illade L, Hernandez-Marques C, Cormenzana M, et al. [Wilms' tumour: A review of 15 years recent experience][J]. An Pediatr(Engl Ed), 2018, 88(3): 140-149.
[3] 杨文萍, 武海燕, 张文, 等. 儿童肾母细胞瘤病理诊断共识[J]. 中华病理学杂志, 2017, 46(3): 149-154. doi: 10.3760/cma.j.issn.0529-5807.2017.03.002
[4] Dome JS, Graf N, Geller JI, et al. Advances in Wilms Tumor Treatment and Biology: Progress Through International Collaboration[J]. J Clin Oncol, 2015, 33(27): 2999-3007. doi: 10.1200/JCO.2015.62.1888
[5] D'Angio GJ, Evans AE, Breslow N, et al. The treatment of Wilms' tumor: Results of the national Wilms' tumor study[J]. Cancer, 1976, 38(2): 633-646. doi: 10.1002/1097-0142(197608)38:2<633::AID-CNCR2820380203>3.0.CO;2-S
[6] Hol JA, Lopez-Yurda MI, Van Tinteren H, et al. Prognostic significance of age in 5631 patients with Wilms tumour prospectively registered in International Society of Paediatric Oncology(SIOP)93-01 and 2001[J]. PLoS One, 2019, 14(8): e0221373. doi: 10.1371/journal.pone.0221373
[7] Aronson DC, Hadley GP. Age is not a prognostic factor in children with Wilms tumor beyond stage Ⅰ in Africa[J]. Pediatr Blood Cancer, 2014, 61(6): 987-989. doi: 10.1002/pbc.24948
[8] 中华医学会小儿外科学分会泌尿外科学组. 儿童肾母细胞瘤诊疗专家共识[J]. 中华小儿外科杂志, 2020, 41(7): 585-590. doi: 10.3760/cma.j.cn421158-20190805-00479
[9] Koshinaga T, Takimoto T, Oue T, et al. Outcome of renal tumors registered in Japan Wilms Tumor Study-2(JWiTS-2): A report from the Japan Children's Cancer Group(JCCG)[J]. Pediatr Blood Cancer, 2018, 65(7): e27056. doi: 10.1002/pbc.27056
[10] Green DM, Breslow NE, Beckwith JB, et al. Comparison between single-dose and divided-dose administration of dactinomycin and doxorubicin for patients with Wilms' tumor: a report from the National Wilms' Tumor Study Group[J]. J Clin Oncol, 1998, 16(1): 237-45. doi: 10.1200/JCO.1998.16.1.237
[11] Gratias EJ, Dome JS, Jennings LJ, et al. Association of Chromosome 1q Gain With Inferior Survival in Favorable-Histology Wilms Tumor: A Report From the Children's Oncology Group[J]. J Clin Oncol, 2016, 34(26): 3189-3194. doi: 10.1200/JCO.2015.66.1140
[12] Ehrlich PF, Ferrer FA, Ritchey ML, et al. Hepatic metastasis at diagnosis in patients with Wilms tumor is not an independent adverse prognostic factor for stage Ⅳ Wilms tumor: a report from the Children's Oncology Group/National Wilms Tumor Study Group[J]. Ann Surg, 2009, 250(4): 642-648. doi: 10.1097/SLA.0b013e3181b76f20
[13] Dix DB, Seibel NL, Chi YY, et al. Treatment of Stage Ⅳ Favorable Histology Wilms Tumor With Lung Metastases: A Report From the Children's Oncology Group AREN0533 Study[J]. J Clin Oncol, 2018, 36(16): 1564-1570. doi: 10.1200/JCO.2017.77.1931
[14] Dome JS, Cotton CA, Perlman EJ, et al. Treatment of anaplastic histology Wilms' tumor: results from the fifth National Wilms' Tumor Study[J]. J Clin Oncol, 2006, 24(15): 2352-2358. doi: 10.1200/JCO.2005.04.7852
[15] HoneymanJN, RichBS, McEvoyMP, et al. Factors associated with relapse and survival in Wilms tumor: a multivariate analysis[J]. J Pediatr Surg, 2012, 47(6): 1228-1233. doi: 10.1016/j.jpedsurg.2012.03.030
[16] Berrebi D, Leclerc J, Schleiermacher G, et al. High cyclin E staining index in blastemal, stromal or epithelial cells is correlated with tumor aggressiveness in patients with nephroblastoma[J]. PLoS One, 2008, 3(5): e2216. doi: 10.1371/journal.pone.0002216
[17] 马成泉, 周智恩, 张震宇, 等. Ki-67指数对肾上腺皮质癌术后的预后评估价值[J]. 临床泌尿外科杂志, 2022, 37(4): 257-260. https://lcmw.whuhzzs.com/article/doi/10.13201/j.issn.1001-1420.2022.04.003
[18] Krishna OH, Kayla G, Abdul Aleem M, et al. Immunohistochemical Expression of Ki67 and p53 in Wilms Tumor and Its Relationship with Tumor Histology and Stage at Presentation[J]. Patholog Res Int, 2016, 2016: 6123951.
[19] Gratias EJ, Dome JS, Jennings LJ, et al. Association of Chromosome 1q Gain With Inferior Survival in Favorable-Histology Wilms Tumor: A Report From the Children's Oncology Group[J]. J Clin Oncol, 2016, 34(26): 3189-3194. doi: 10.1200/JCO.2015.66.1140
-

| 引用本文: | 储婧. 儿童肾母细胞瘤临床病理资料与预后的影响因素分析[J]. 临床泌尿外科杂志, 2023, 38(4): 260-264. doi: 10.13201/j.issn.1001-1420.2023.04.005 |
| Citation: | CHU Jing. Clinicopathological data and prognostic factors of nephroblastoma in children[J]. J Clin Urol, 2023, 38(4): 260-264. doi: 10.13201/j.issn.1001-1420.2023.04.005 |
- Figure 1.
- Figure 2.
- Figure 3.
- Figure 4.



 下载:
下载: